|
|
|
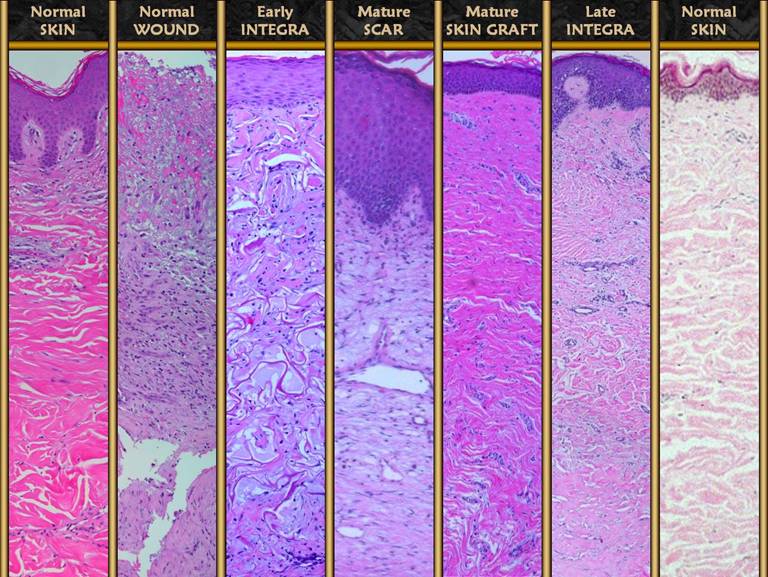
SLIDE 1 Histogenesis versus Wound Repair: the Anatomy of Integra’s Properties
Marc
E. Gottlieb, MD, FACS Arimedica version, 2005. Originally presented at: The Boswick Burn and Wound Meeting, Maui,
February, 2003. Subsequent presentations, 2003 – 2005. Copyright © 2003 - 2006, Marc E. Gottlieb, MD Content may be used for non-commercial
educational purposes. Content may not be republished, nor used for
commercial purposes without prior license or permission, except as permitted
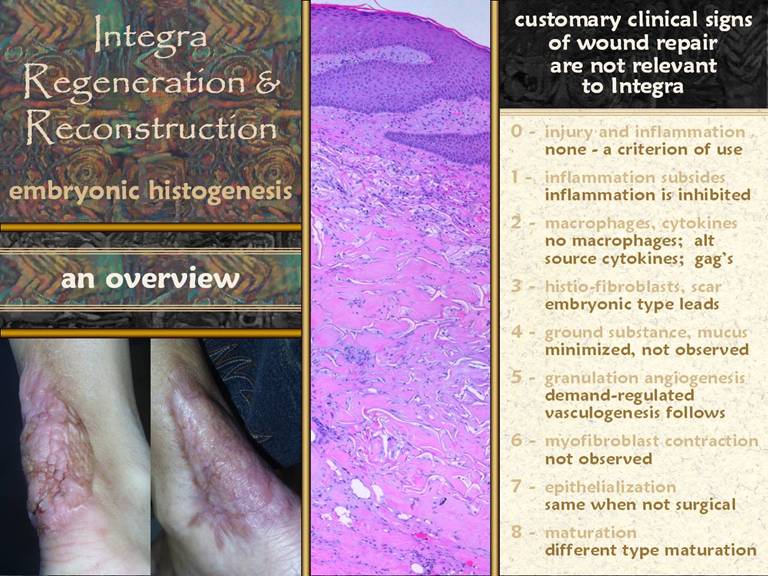
as “fair use” under United States copyright laws. Contacts: Marc E. Gottlieb, MD, FACS Plastic Surgeon, Phoenix, Arizona Phone: 602-252-3354 Fax: 602-254-7891 Email: megott@arimedica.com Introduction. Integra is a semibiological device used as a
skin regeneration matrix. Among other
desirable properties, it controls or prevents scar. In reconstructive and burn surgery, it
gives superior results with fewer sequelae compared to split thickness skin
grafts. This superiority derives from it’s ability to (1) suppress the normal response to
injury, the process of inflammation and conventional wound repair, and (2) to
instead induce a state of histogenesis comparable to embryonic
processes. The similarities of Integra
and embryonic histogenesis, and their distinction from inflammatory wound
repair, can be inferred from prior knowledge about the cell and system
dynamics of growth, repair, and regeneration.
In this report, histological examination by high resolution light
photomicrography corroborates these hypotheses. Inflammatory wound repair. Injury is recognized by platelets. Inflammation is the auto-amplifying
response that defends the host, mediated first by platelet releasates and
then by the blood borne leukocytes which they attract. Mononuclear leukocytes are transformed into
macrophages. As inflammation subsides,
macrophages, via cytokines, orchestrate repair by marshalling cells from
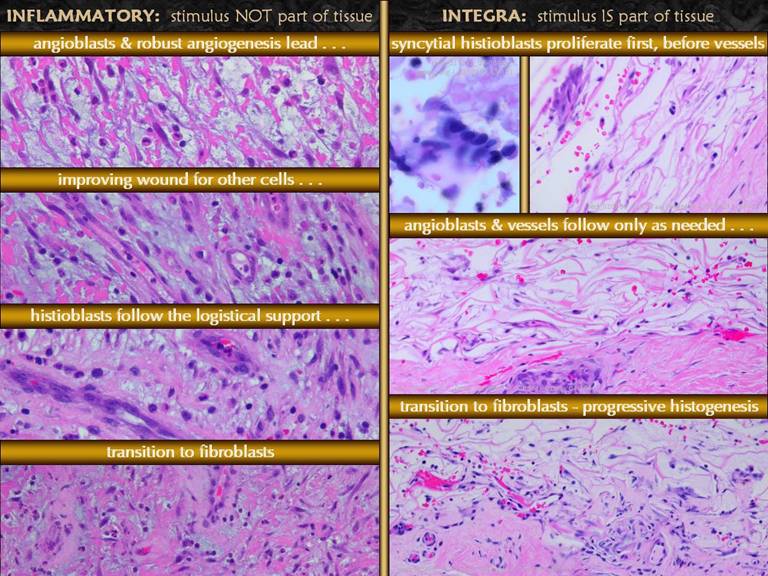
three local stem lines: endothelium, histioblasts, and epithelium. In an integrated process called the “wound
module”, endothelial angiogenesis restores environment, allowing histioblasts
and their progeny, fibroblasts and myofibroblasts, to contract and repair the
wound with connective proteins.
“Closure” of the wound is the sequestration of mesenchymal cells and
tissue from the ambient world by the restoration of a continuous
epithelium. These events,
inflammation-subsidence, macrophages, angio-genesis, fibroplasia, and
epithelialization, occur in that order, since each subsequent stage is
dependent on its predecessors. These
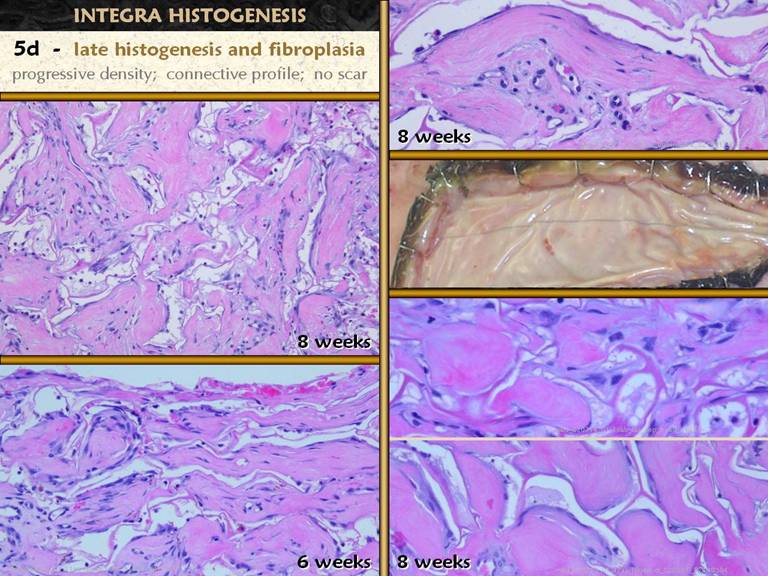
events are observable in any open wound as it heals. Integra and histogenesis. Integra is a spongy material of type 1
collagen and chondroitin-6-sulfate.
Applied to a fresh wound, recognition of injury is halted. This results from the chondroitin which
masks the platelet binding sites on the collagen (unlike normal collagen and
its products, Integra is non-thrombogenic).
By inhibiting platelet adhesion, the process of inflammation, macrophages,
and the macrophage-dependent wound module is entirely arrested. Glycosaminoglycans, the basis of
extracellular ground substance, regulate embryonic wound repair, which is
simply a process of continued histogenesis rather than inflammation and fibroplasia. The chondroitin in Integra is thought to be
the signal which tells cells from the adjacent host wound to initiate a
process of embryonic histogenesis. To
cells in the host tissue, because there is no inflammation, they do not
recognize the wound as such. They
simply see an empty scaffold of pseudo-ground substance, devoid of cells, and
they wander into the scaffold to begin the process of creating tissue. Histology. Integra remains devoid of acute
inflammatory cells. Within days, it
begins to be slowly and sparsely populated by migratory angiopericytes. Once in the matrix, these cells transform,
spawning fibroblasts with nuclei and cytoplasm enlarged for
proteogenesis. Abundant amorphous
collagen appears. Vasculogenesis of
the embryonic type occurs by angioblasts responding only to areas of active
fibroplasia. Later, collagen starts to
organize and mature, eventually looking more like normal dermis rather than
scar. Formation of a papillary dermis
occurs in response to epithelial closure (which occurs surgically). Summary. Normal inflammatory wound repair is an
amplified or open loop process which makes an abundant excess of repair
tissue which is then resorbed and thinned during the late phase of wound and
scar maturation. In distinction,
Integra histogenesis is a controlled process which slowly and incrementally
builds a model of normal tissue which does not need to undergo
resorption. Integra histogenesis is an
altogether different process than wound repair, and the differences account
for Integra’s desirable properties.
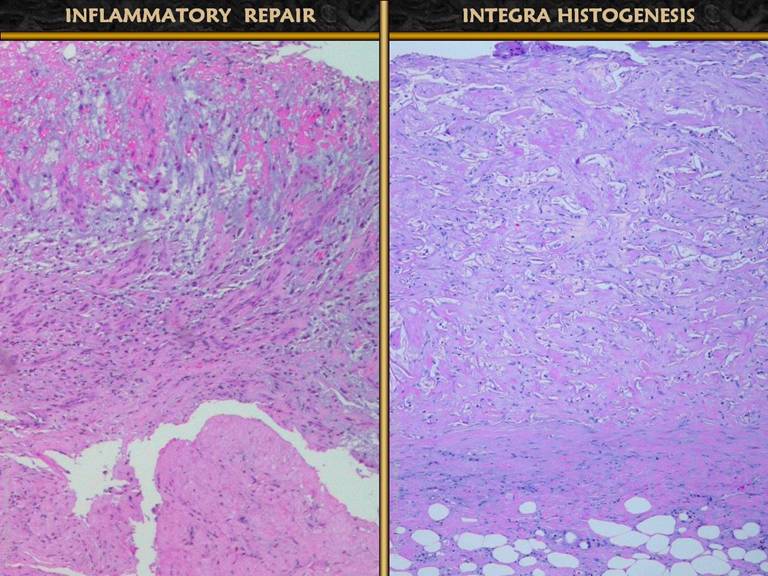
Side by side photographic comparison of wound histology, inflammatory
wound repair versus Integra histogenesis, confirms these physiological
processes and their distinct differences. |
PART 1: NORMAL INFLAMMATORY WOUND REPAIR
|
|
|
|
|
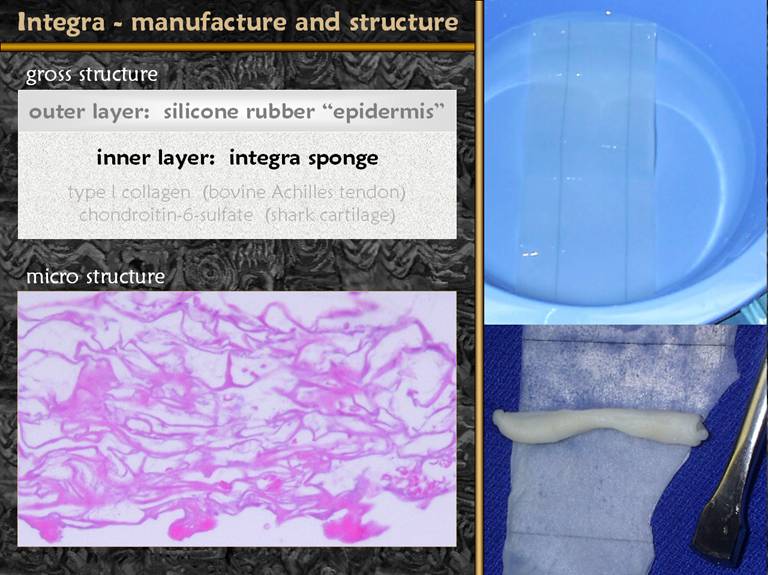
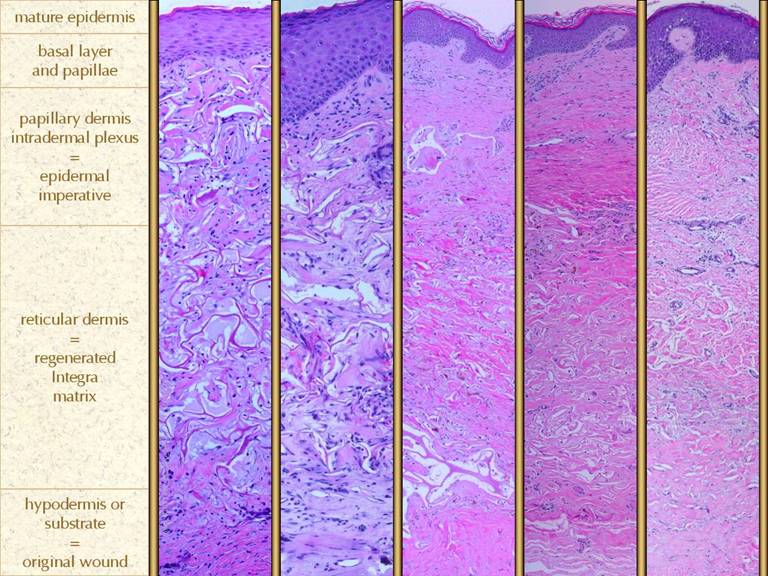
SLIDE 2Integra (aka Integra Artificial Skin) is a bilayer sheet. The spongy bottom layer (microscopic view shown) is made of Type I collagen (from bovine achilles tendon) and chondroitin-6-sulfate (made from shark cartilage). The reticulum or mesh size of the sponge is engineered to mimic the size of the connective tissue reticulum in normal human dermis. |
|
|
|
|
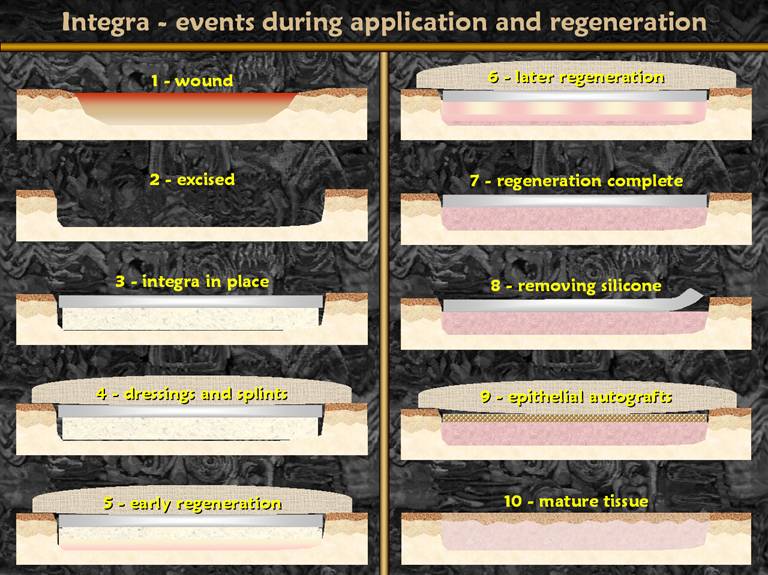
SLIDE 3Integra is used in the following way. A wound or defect is first cared for until the wound is clean and meets criteria for closure. During surgery, the wound surface is completely excise. Integra is put in place and secured with compression and fixation dressings. Early regeneration of tissue within the sponge starts adjacent to the host wound. As time goes by, advancing histogenesis fills the sponge with an analogue of normal dermis. The progress of regeneration can be observed directly through the silicone outer layer. When regeneration is complete, averaging about 4 weeks in wounds and people, the silicone is removed and skin grafts are placed on the regenerated lamina, thereby completing the skin reconstruction. |
|
|
|
|
SLIDE 4Integra is of proven value in managing burns, fasciitis,
chronic wounds, and problematic reconstructions. Its benefits, good results, utilitarian
usage, and superiority to older or conventional reconstructive and wound
closure options is understood. Some of Integra’s special properties and
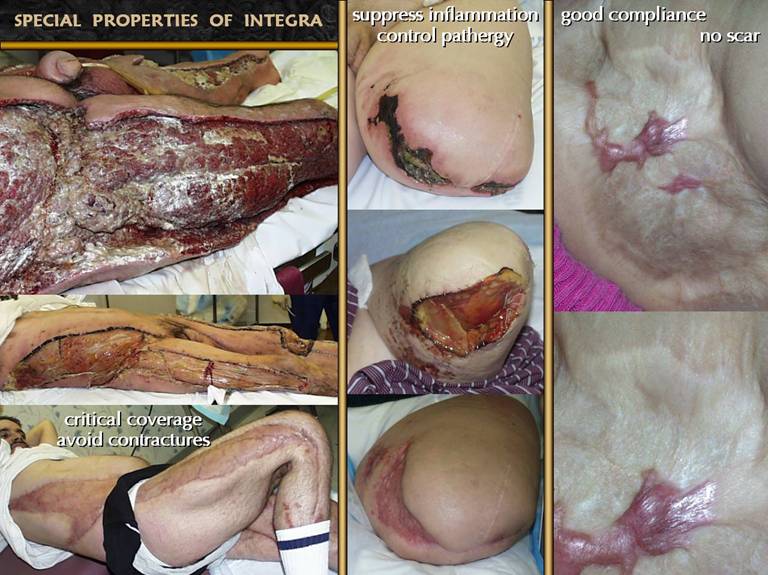
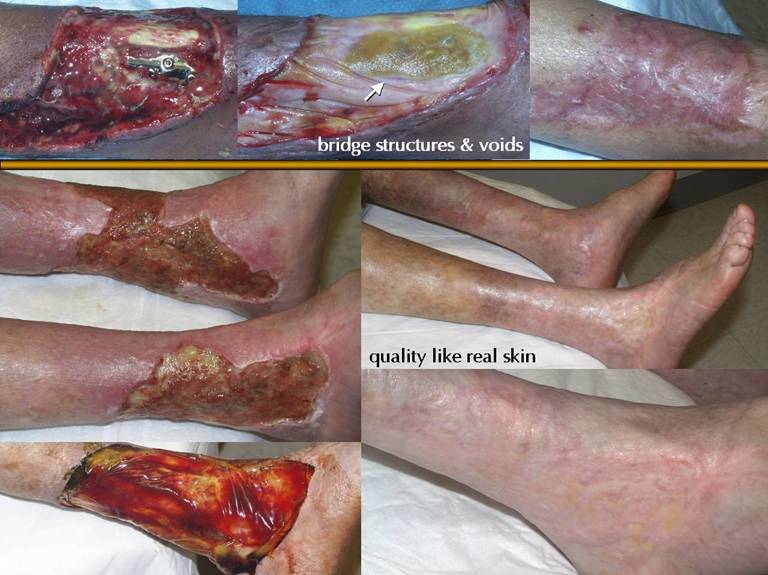
related outcomes are shown on these two slides. Left. Critical
wound closure. Absence of
contractures. This patient had Group A
streptococcal necrotizing fasciitis.
At 8 days, the wounds are clean, well bathed, and packed in silver
sulfadiazine. Nevertheless, bacterial
transients in these healthy wounds, plus a white blood cell count of only 200
due to bone marrow failure made him continuously septic with imminent
death. The patient had a complete
turnaround of septic physiology immediately upon placing Integra. No late reconstructive procedures were
needed because there were no contractures. Middle. Suppress
inflammation. Control pathergy. This patient has severe atherosclerotic
occlusive disease. Toe, then foot,
then leg, then thigh amputations were done, each with progressive skin and
fascia necrosis. Debridement and
immediate closure with Integra avoids further necrosis. In marginal wounds, with severely limited
degrees of freedom within the machinery of repair, exhibiting quenched
chaotic dynamics, injury is subject to pathergy and progressive
complications. By providing high
quality coverage to the wound, by suppressing inflammation, pathergy and
complications are arrested. Right. Good compliance. No scar. Integra can control or avoid contractures, keloids, and other scar problems, because what it regenerates, an analogue of normal dermis, is distinctly unlike scar. In these pictures, a small area of Integra did not take, leading to normal inflammatory wound healing and hypertrophic scar. The Integra skin is not strictly normal. There are pigment irregularities (as for any skin graft), there are no subcutaneous fascias (they were removed during disease and surgery), and the texture is somewhat irregular. Nevertheless, the wrinkles, fine folds, mature appearance, and other signs of soft pliable compliant skin stand in sharp contrast to the young active scar at the middle.
|
|
|
|
|
SLIDE 5Special properties and outcomes, continued. Top. The patient
had an ankle fracture, fixation, wound necrosis, then necrosis of multiple
free flaps. The wound was cleaned up, then Integra was applied in its role as “artificial skin”. The plan was to place new Integra every
four weeks, as each previous piece regenerated and the silicone was about to
be ejected. This way, Integra would be
the interim skin until the fracture was healed and the plate could be
removed. However, the ability of
histogenesis to advance through the sponge meant that new tissue formed over
the plate. Advancing tangential
histogenesis is apparent at the margins (arrow). After three pieces of Integra, the plate
and tendons were closed by a regenerated lamina of new tissue. The plate, fracture, and reconstructed skin
have all healed and remained stable and problem free for one year. Bottom. Leg ulcers of forty years duration in a patient with Sjögren’s syndrome. Fasciectomy and skin reconstruction leads to healed wounds, without any joint contractures nor hypertrophic scars. The reconstructed skin looks surprisingly normal, and not at all like conventional skin grafts would have looked. |
|
|
|
|
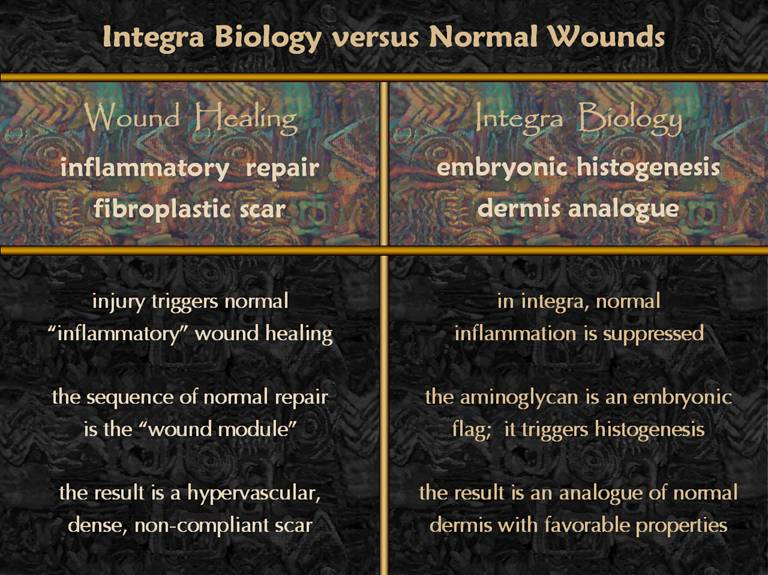
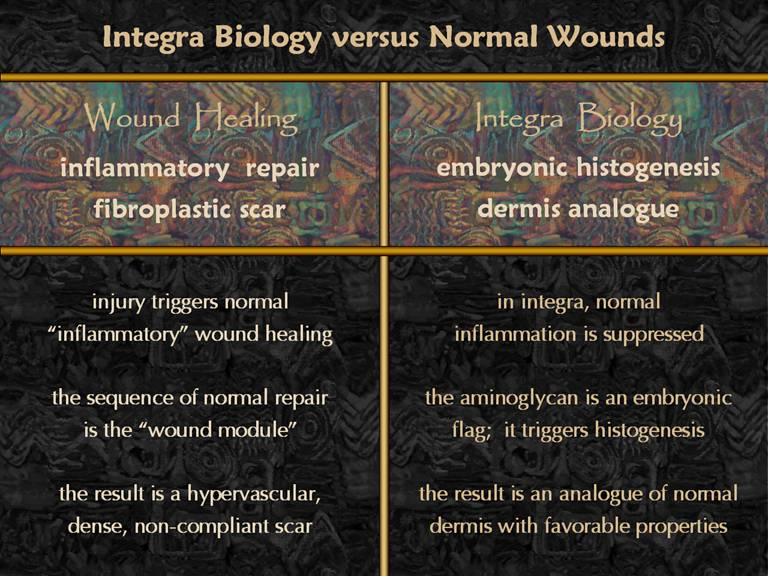
SLIDE 6Integra has special properties and superior results
compared to skin grafts and other scars.
Why? The answer is that Integra
regeneration, which is akin to embryonic histogenesis, is distinctly
different than normal wound healing, the ordinary programmed response to
injury and inflammation. Normal inflammatory
wound healing follows a pattern called the “wound module” which glues the
injury together with fibroplastic scar, scar being that undesirable material
which is thick, non-compliant, unsightly, and prone to problems. Integra actually suppresses inflammation
and the wound module. The aminoglycan
in the Integra is a signal to the body of embryonic conditions, triggering a
process of normal histogenesis, leading to more normal tissue. This presentation will first show the histology of normal inflammatory wound repair, then the histology of Integra histogenesis, and then compare the two. This will demonstrate that there is a cellular basis for the difference between the two, and that Integra’s desirable and distinctive properties have an understandable basis. |
|
|
|
|
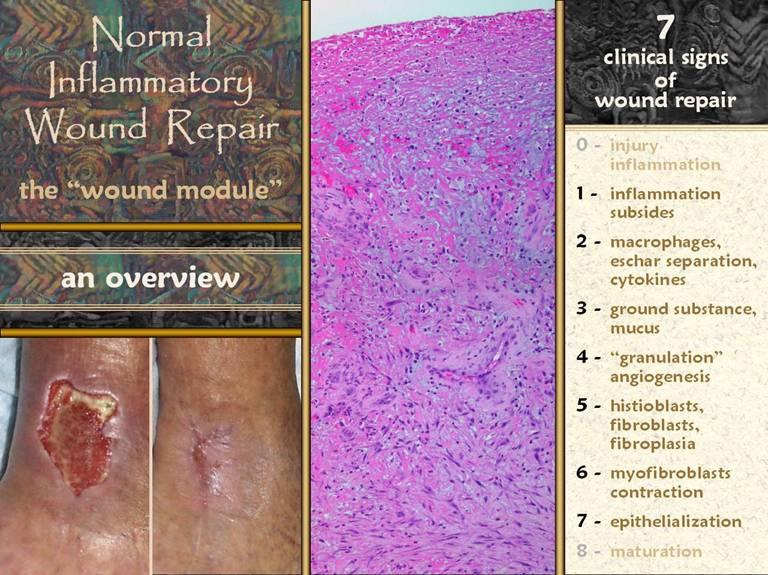
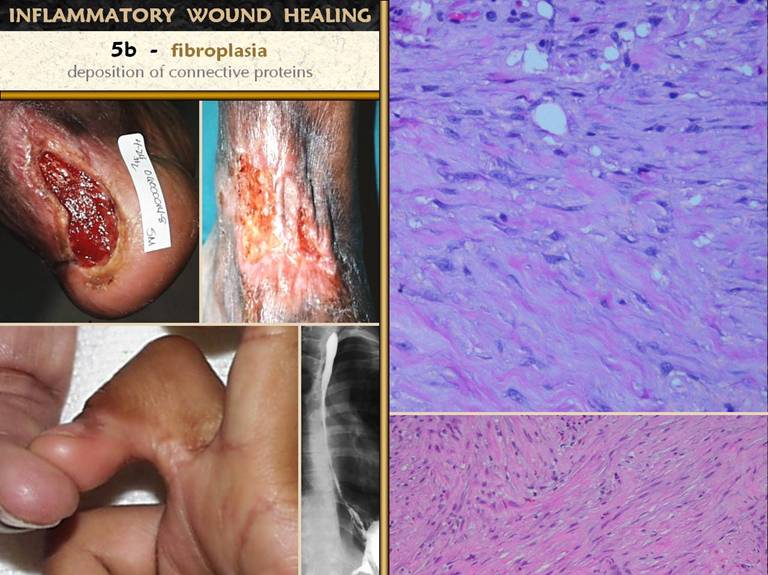
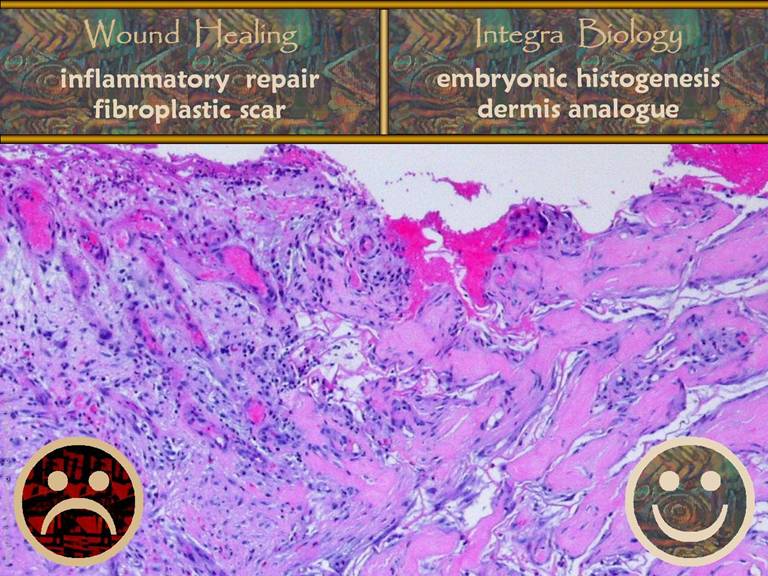
SLIDE 7The picture shows a wound - open and then healed, by
normal physiological processes. The
microscopic picture shows what is happening in such a wound. This is the “wound module”, the normal
process of inflammatory wound repair.
The major physiological events and accompanying clinical signs are
listed. Injury and inflammation must
be controlled for adequate repair to begin.
After the wound is closed - fully re-epithelialized - the nominal
clinical endpoint of complete repair, then the wound matures. In between, there are 7 notable and
clinically observable events: 1 -
inflammation subsides; 2 - macrophages
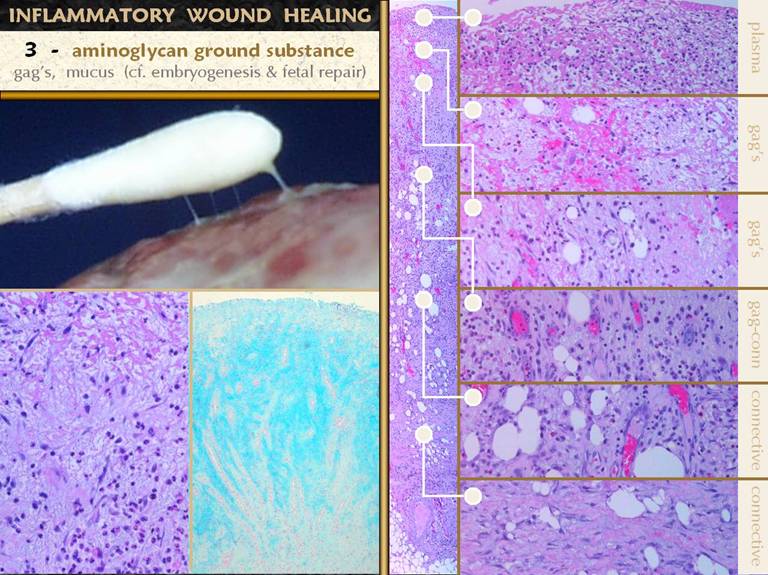
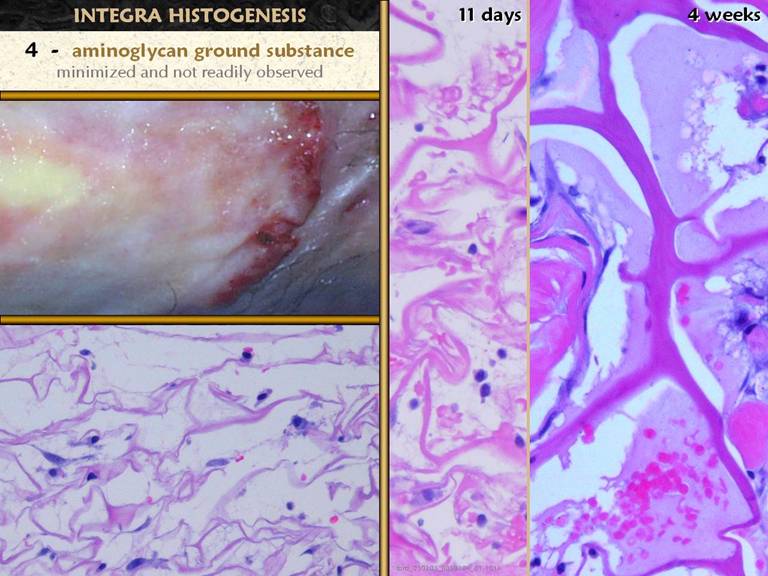
appear, separating eschar, and orchestrating local cells by cytokines; 3 - aminoglycan ground substance
appears; 4 - angiogenesis occurs,
visible as “granulation tissue”; 5 -
histioblasts appear, leading to fibroblasts, which make connective proteins
to hold the wound together; 6 -
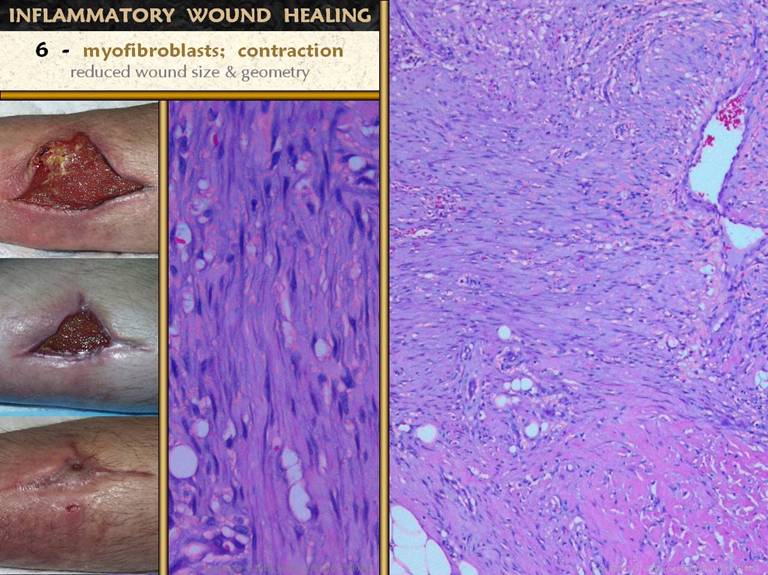
myofibroblasts are another histioblast derivative, which serve to contract
the wound, responsible for much of the wound closure; 7 - epithelial growth continues until there
is a complete epithelial (ectodermal or entodermal) interface between the
environment and the mesenchyme.
An Important Note: The wound repair process develops in time. If the wound is open, newer inflammation and wound repair accumulates on the surface. The net effect is that the deeper you look down from the surface, the older in time you are looking. When you look at the histology of normal inflammatory repair, and as you look at the photographs here, remember that each specimen shows its own history. At the surface are events occurring now. As you go deeper, you are seeing, in sequence, events that happened yesterday, the day before, the day before that, and weeks before. Changes occur more slowly deeper down, with less accumulation of depth, so if you plot depth(y) versus time(x), you get a logarithmic type curve. The way the wound looks under the microscope did not happen all at once. However, most of the images shown here are chronic wounds, so each image captures the whole history of the wound. |
|
|
|
|
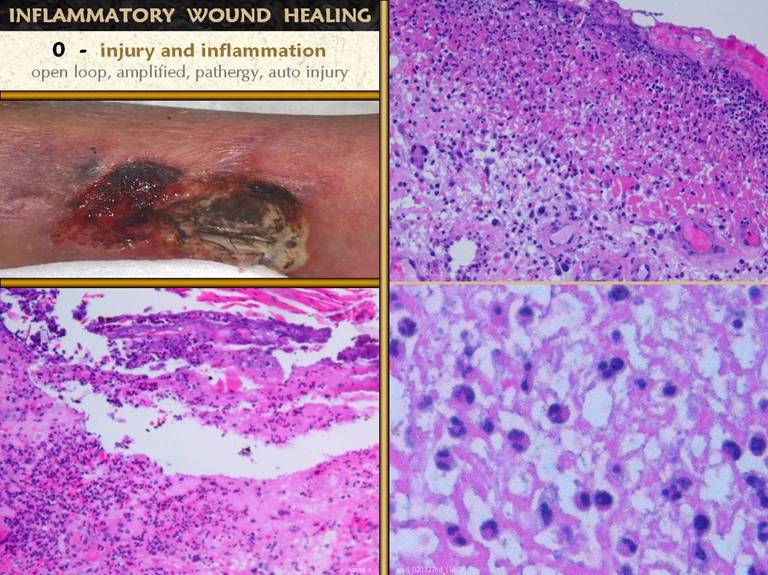
SLIDE 8Injury, by any means, is what triggers the process of
inflammation and repair. Inflammation
is the system for recognizing and responding to an injury, the means of
defending the host, and the means of preparing for repair. It is in many ways either open loop or
auto-amplifying. Once triggered, the
response is dramatic and intense.
While meant to contain, control, and damage pathogens, its inherently
destructive nature can also damage the host.
In the sick host, with underlying disease and risk factors and limited
degrees of freedom in the wound, inflammation is the cause of pathergy -
paradoxical death and destruction of host tissues. The ankle ulcer is in a patient with severe
uncontrolled Behçet’s syndrome, where any minor cut, scrape, or bruise,
including debridement and biopsy, can cause progressive destruction. Histologic features of acute inflammation
include: Lower left. A small
ulcer in a patient with immune vasculitis.
Infarcted epidermis and superficial dermis are separating from deeper
structures. The entire area is filled
with small acute inflammatory cells, mostly polymorphonuclear leukocytes. Upper right. The
upper layer of any typical wound, due to benign trauma or ulcerative
pathology or anything else.There is a very dense infiltrate of poly’s,
lymphocytes, and monocytes (“acute inflammation”) in a pink eosinophilic zone
composed of plasma proteins and fluid exudates. Deep to this is a more basophilic or
non-staining area where the tissue is sheltered from the ambient environment
and the process of repair can begin, confirming that the inflammatory
infiltrate is doing its job of host defense. Lower right. A close up view of the acute inflammatory infiltrate in the upper zone. Notice the mix of cell types, some mononuclear, but mostly poly’s, more or less in proportion to their numbers in circulating blood from which they are all derived. |
|
|
|
|
SLIDE 9For the wound to begin healing, inflammation must be under
control. As seen on the previous
slide, healing can begin in the zone deep to the surface inflammation,
assuming that the inflammatory layer is satisfactorily sequestering
environment and injury from tissues underneath. The more intense the injury, the deeper the
inflammation, and the less likely is repair to occur. With good clinical care, the cause of
injury and inflammation can be minimized or eliminated, and inflammatory
infiltrates subside, as shown here.
Resolution of acute inflammatory signs and symptoms is the clinical
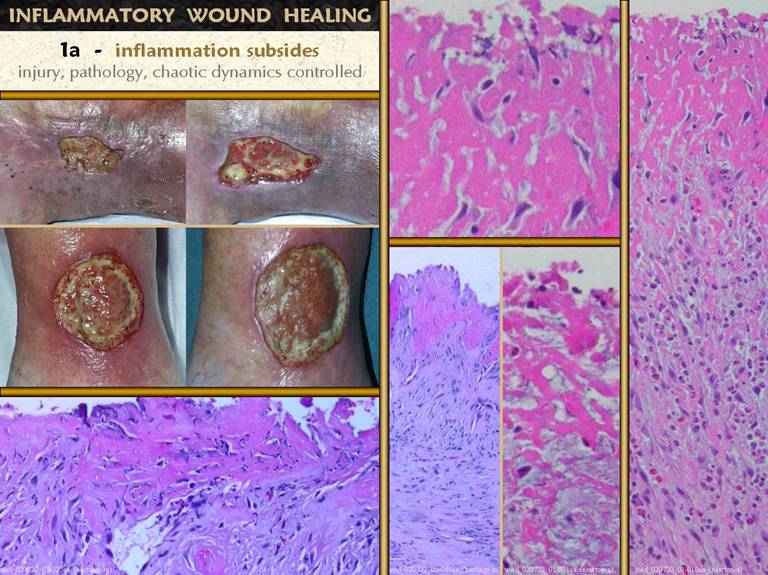
marker that wound repair can begin or has begun and can continue. 1 - top left. Two wounds illustrating the effects of good
care to control inflammation and injury in a wound. Robust active wound repair cannot fully
commence until injury, pathology, inflammation, and chaotic dynamics are
controlled. As the gross signs of
inflammation subside, so do the histologic markers of such, as seen in the
other panels. 2 - left and top, wide and
close-up views. This is a chronic
pressure ulcer that has been closely managed, including twice daily good soap
and water hygiene and topical silver sulfadiazine. The upper exudate layer is devoid of acute
inflammatory cells. Large transformed
macrophages are in this layer, and below this the proliferative reaction to
their stimulation is beginning. 3 - bottom center, right. Another view of a wound well cared
for. This is a wound, and therefore
has nominal degrees of inflammation, but the number of neutrophils is scant. 4 - bottom center, left . This open wound likewise has continuing
care with good hygiene and silver sulfadiazine. Acute inflammatory cells are scant or
absent, and fibrous proliferation is present at shallow depths below the
surface. 5 - bottom left. A close up showing more mature wound module angiogenesis very close to the surface, without a significant exudate-inflammatory layer. |
|
|
|
|
SLIDE 10In the process of normal inflammatory wound repair,
inflammation and repair are a tightly integrated process in which repair is
triggered by inflammation. This is
true, even if the wound remains closed.
The leg above in a patient with chronic venous disease has dense
liposclerosis and obliteration of subcutaneous fascias by scar. Even though the skin was never ulcerated,
the repetitive cycles of venous vasculitis and stasis dermatofasciitis lead
to the wound module and fibrosis. How
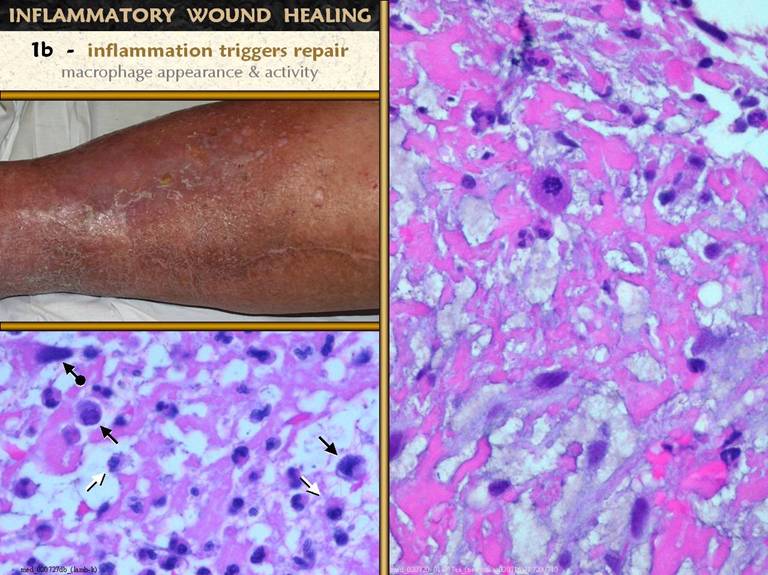
does inflammation trigger repair? Below. Neutrophils
and lymphocytes have a host defense function.
They do their job then die or clear out. Monocytes are the key. They are the root of repair. Under the influence of transforming
cytokines from platelets, leukocytes, and other inflammatory triggers, they
begin to enlarge, accumulating cytoplasm and
nucleoplasm, then they become amoeboid.
The white arrows show monocytes that arrived at the top of the wound
with the normal influx of inflammatory leukocytes. The black arrows show enlarging, transforming
monocytes. The arrow-and-circle is a
cell whose transformation from monocyte to macrophage is complete. Right. The wound surface has some inflammatory cells. Below this is the transition zone with transforming cells. At the bottom of the field are numerous macrophages. These macrophages initiate and orchestrate the rest of the repair process. Go back to the previous slide and look for these features on those specimens. |
|
|
|
|
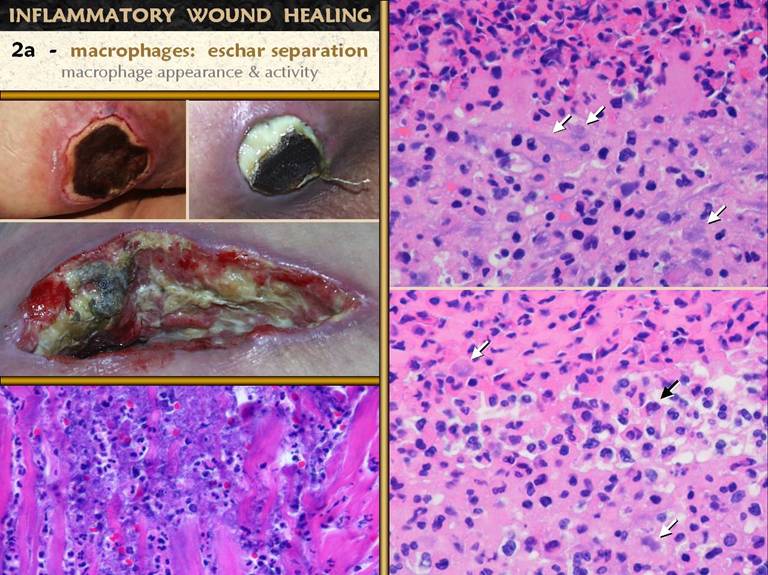
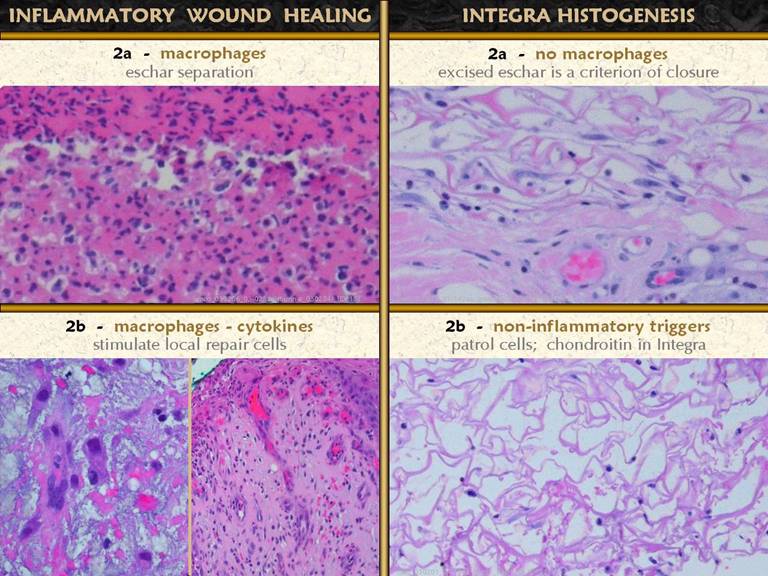
SLIDE 11Macrophages (those transformed monocytes) have two
functions on the wound. The first is
as a phagocyte. They can discriminate
healthy tissue from debris, and they start to clear out the debris. Clinically, this is seen as eschar
separation. The wound photos show
eschar in varying phases of evolution and separation. Bottom left. The
interface between necrosis and living tissue at the edge of a pressure
ulcer. Top, basophilic, necrosis. Bottom, eosinophilic, alive. there is an
intense acute inflammatory infiltrate.
Paler larger macrophages are scattered through the interface. Upper right. This
is seen closer up in another patient and wound, also benign pressure
ulceration. The upper half is
necrotic, with lots of inflammatory cells and cell debris. The lower half appears more basophilic than
the necrosis, not because it is necrotic, but because of large pale
macrophages at the interface, interspersed with other smaller inflammatory
cells. Bottom right. Same orientation as above. The central third is the cleavage zone. This is where eschar is separating from remaining living tissue. |
|
|
|
|
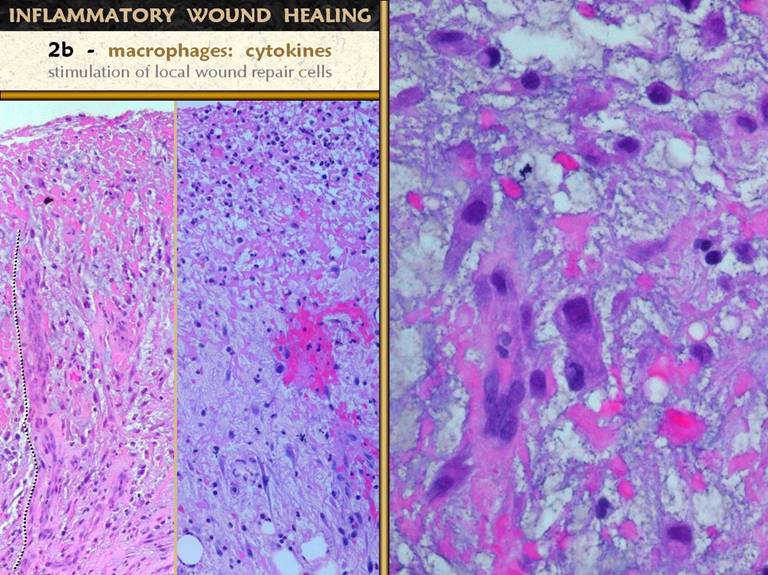
SLIDE 12As discussed above, macrophages initiate and orchestrate
the repair process. They do this by
making numerous cytokines which stimulate cells. The monocyte-macrophages are blood borne,
but the cells which do the work of repair are local. Two cell lines must be triggered,
angiogenic cells and histioblasts. Left and center: A
zoomed out view showing the upper inflammatory zone, the subjacent zone of
macrophage transformation, and below that the zone of angioblast
streaming. On the left, the
angiogenesis is more mature, with cells mostly coalesced into new
blood-conducting vessels nearly all the way up to the inflammatory zone (one
such vessel is traced with a dotted line alongside). In the center picture, streaming
angioblasts are in abundance, but not many vessels (only the one along the bottom
right corner). Right. A close up view near the top of the wound. At the top, monocytes are transforming, and below, they are mature macrophages. The organized cluster of cells is an angiogenic cord. These angiocytes have zoomed up from vessels below, aiming directly at the source of chemotactic stimulation, the angiogenic cytokines made by the macrophages.
|
|
|
|
|
SLIDE 13The ground substance that all cells float in is made
largely of glycosaminoglycans (GAG’s).
Many mature tissues, with dense cellular parenchymas or thick fibrous
stromas have little ground substance.
Some tissues, notably embryonic ones that have little connective
protein, stroma depend on the GAG’s to be the substance of form and organization,
the creamy pudding for their pearls of tapioca. This is true in the sub-inflammatory layer
of the wound. In the surface layer of
acute inflammation, there is a plasma-like layer of proteinaceous exudates. Below this, in the zone of
monocyte-macrophage transformation, and below that in the zone of
angiogenesis, these living cells depend on GAG soup to have a hospitable
environment. A fibrous stroma, made by
fibroblasts comes deeper in the wound, because angiogenic new vessels must be
in place before histioblasts and fibroblasts can function. Mucus exudates are easily recognized
clinically on the wound surface. These general zones are shown above, right: 1 - top layer,
proteinaceous, inflammation; 2 -
monocyte-macrophage transformation and cytokine release, GAG’s; 3 - angiocyte streaming and loose
angiogenic organization, GAG’s; 4 -
organized vessels, histioblast proliferation, GAG’s and early unorganized
connective proteins; 5 - histioblasts
becoming young fibroblasts, fibrous stroma fills most of the space; 6 - mature fibroblasts with dense collagen
and lamellar organization. Left, another hematoxylin-and-eosin view of the upper wound layers, showing a loosely organized tissue, with cells able to wander freely, with no fibrosis. This is the glycosaminoglycan environment of the upper wound. While the H&E histology allows the location of the aminoglycans to be inferred, the Alcian blue stain shows the tissue GAG’s (it stains carboxylated and sulfated aminoglycans such chondroitin, hyaluronan, dermatan). The dense blue stain is present in the sub-inflammatory macrophage layer, the streaming angioblast layer, and the vessel organization layer. Deeper down, in the zone of fibroplasia, the aminoglycans are less dense, and counter-stained cells are more dense. The origin of the GAG’s is presumably the macrophages and inflammatory cells. |
|
|
|
|
SLIDE 14So far in the acute wound, acute inflammation has
protected the host and stabilized the injury and established a crop of
monocytes-turned-macrophages. GAG’s
have accumulated, creating a substance in which migratory cells can
proliferate and establish organized structure. The macrophages issue cytokines which
muster local cells to create the tissue of repair. Angioblasts-angiogenesis and
histioblasts-fibroplasia are the two main events which glue the wound
together and create a foundation for epithelial growth and wound
closure. The macrophages are knocking
on the door for angioblasts and histioblasts.
Who is answering? Left. Four images
of normal blood vessels, taken of tissues biopsied from clean healthy acute
wounds following excision of one thing or another. These views show thinner and thicker
vessels, larger and smaller, tangential, longitudinal, transverse, through
the lumen or on the surface. These
vessels are made of normal angiocytes.
Cells are flat, thin, cylinderized around the lumen. Endothelial cells are flat. Note that these are all small vessels,
capillaries and arterioles and venules.
Large vessels with a muscular media and elastic lamina are not
shown. Yet these vessels, except for
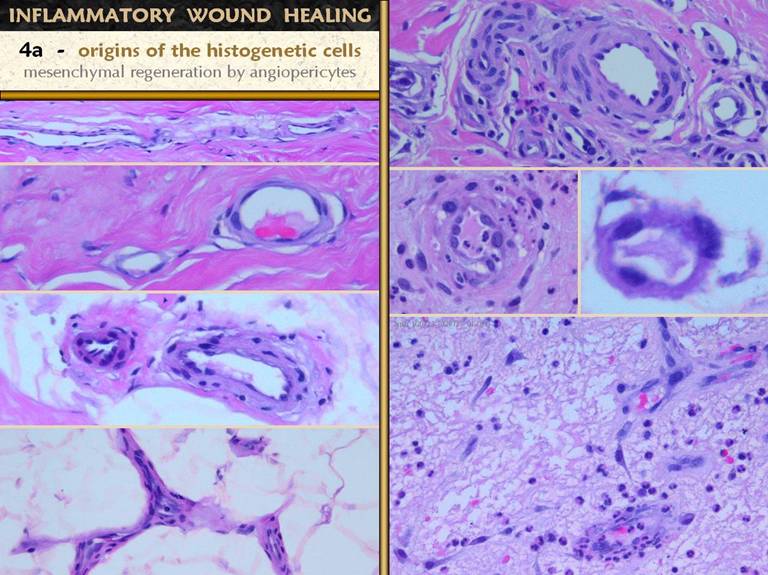
the smallest capillaries, have more than just one layer of cells. The onion-skin layers of cells around the
central endothelial layer are the vascular pericytes. These angiopericytes are the histogenesis
precursors. Under stimulation by
macrophage cytokines or other suitable stimulus, these cells will “come to
life” to heal the wound. Right. A few days after injury, vascular cells in the wound have become hypertrophied. The angiopericytes are thickened, with larger cell bodies and nuclei. Even the endothelial cells have become larger and rounder and can source primitive cells. Even the smallest capillaries can respond. Look at the images on the next slide. As vessels get older and therefore deeper in the wound, they quickly stop “feeling” the stimulation of macrophages above, and they become well-organized vessels with a return to normal cellular architecture. Angiogenesis at the leading edge of the wound depends on the stimulation of the closest vessels, which means even new ones nearby. Thus the youngest vessels remain hypertrophied and are the source of new angiogenic cells even as they are forming. In the bottom image, very young vessels still have thick primitive cells, and a stream of new angioblasts streaming from them up toward macrophages above is obvious. These are already functional vessels, filled with erythrocytes, and leaking leukocytes. |
|
|
|
|
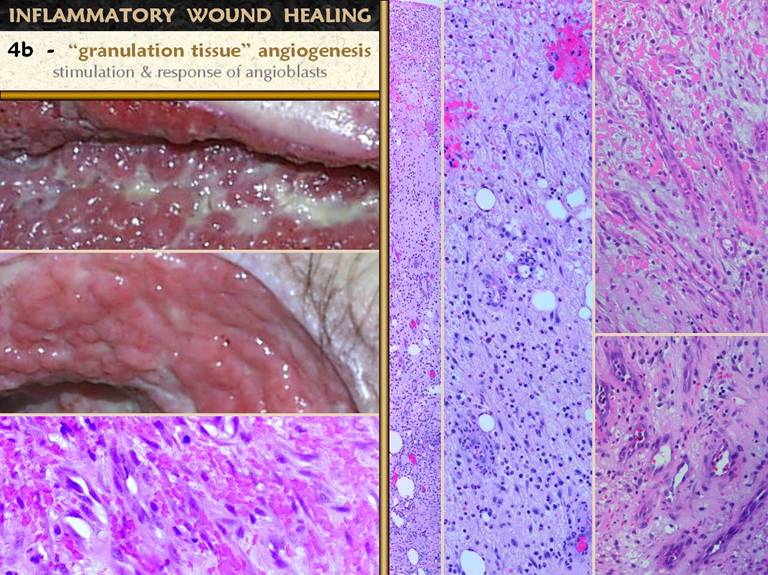
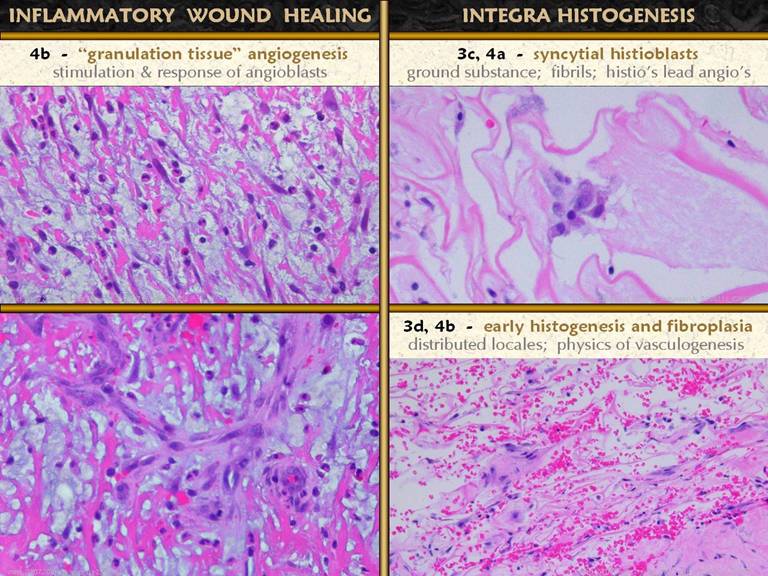
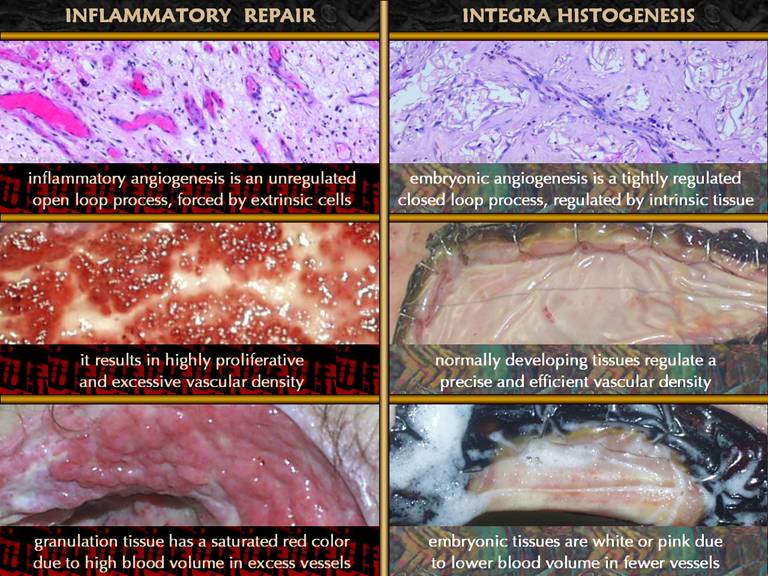
SLIDE 15“Granulation tissue” is the one sign of a healing wound
that the average physician can recognize or label. It is recognizable because of its pink
color, due to blood in proliferative new blood vessels. The proliferation of blood vessels
establishes the crucial supply network that then permits
histioblasts-fibroblasts to flourish and make connective proteins. The angioblasts are cells derived from the angiopericytes
around vessels deeper down. Whether
the angioblasts are different cells or of different origin than the later
histio-fibroblasts versus same-source cells responding according to the
timing and origin of transforming cytokines is unknown. Long vertical images.
Lumens and erythrocytes mark the location of organized new blood
vessels. Right upper.
Streaming angioblasts are highly organized, forming vessels right up
to the sub-inflammatory zone. The vessels
here all show a directional orientation, reaching toward the macrophages that
are stimulating them, coming originally from old established vessels at the
base of the wound, and later on from newer more superficial vessels
established more recently in the life of the wound. Right lower.
Organized vessels deeper in the wound.
The geometry and topology of the vascular network has become more
complex here, as vessels sprout in all directions, to accommodate the needs
of histioblastic cells which are proliferating among the early established
vessels. Note that vessels at this
level are excessive in number compared to the vascular density of normal skin
and fascias, but that the vessels are otherwise fairly mature looking, with a
single well-organized layer of cells that are no longer enlarged or
hypertrophic. Left lower. An example of “granulation tissue” tissue that is densely packed with vessels. The view shows mostly erythrocytes. The basophilic nuclei are all angioblast or young angiocytes. There are no inflammatory cells, no macrophages, and only a few fibroblasts or non-committed histioblasts. |
|
|
|
|
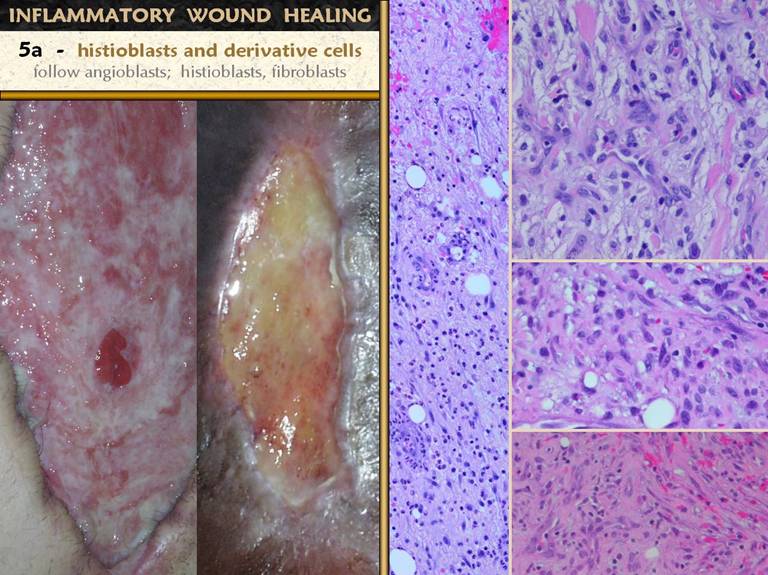
SLIDE 16As angioblasts make vessels and establish an environment
in which later cells can proliferate, many new cells appear which will mature
into fibroblasts and myofibroblasts.
Fibroplasia is not always visible in wounds or wound photos, except as
the final skin scar. In the two
photos, an abdominal wound after trauma and a chronic ankle ulcer, angiogenic
“granulation tissue” is thin, and the deeper layer of fibrosis can be seen. In the microscopic pictures, the upper wound a close-ups are shown. Long image. At the
top is the macrophage transformation zone, and below this the angioblast
streaming zone. Just above the middle
are some organized vessels, and between them are small cells with round
nuclei. These cells become denser and
more numerous going toward the bottom. Upper right. This
image is a different wound than the vertical image, but it corresponds in
depth to the bottom of the long image.
There are organized mature vessels interspersed with the other
cells. These are the
histioblasts. They are starting to
elongate into spindle shapes - fibroblasts.
While the matrix is still largely aminoglycans (non-staining areas),
thin strands of eosinophilic young collagen are starting to appear. Middle right. A
little bit deeper, in another wound.
There are vessels at bottom and upper right, and between them
histioblasts and young spindled fibroblasts are quite dense. More of the space is occupied by pale pink
collagen. Bottom right. In another wound, deeper yet. Histioblasts and young fibroblasts remain dense. the space is almost completely filled by young disorganized collagen. The cells are, in general, less round, more spindled, and starting to take on some organization in the form of stratification or lamellations. |
|
|
|
|
SLIDE 17This slide is a continuation of the previous one. The previous one focused on the appearance
of histio-fibroblasts. This one
focuses on their end product, the fibrous scar. Note that throughout this discussion, while
collagen alone is referenced for convenience, the process involves all of the
connective proteins, such as elastin and fibronectins, which have greater or
lesser roles in this process depending on various circumstances. Top right. Just
below the zones shown on the last slide, the randomly arranged young
fibroblasts start to become flatter and layered. They are stratified between maturing wavy bundles
of collagen. Bottom right. At a
deeper layer again, the stratification and organization of the scar is
obvious. The scar bundles are thick, and different bundles criss-cross in different
directions. Left. Scar is the glue that cements the wound together. While it is crucial to restore the mechanical integrity of the injured part, the dense pack of collagen seen on the right leads to undesirable properties. These are photos of scar complications. 1 - an achilles ulcer that is trying to heal, a competent wound module, but where scar has made the skin edges so non-compliant that they cannot contract. 2 - an anterior ankle burn scar, hypertrophied due to tensile loads (Wolf-Davis Law), resulting in a non-compliant leash that fractures with plantar flexion, triggering more inflammation and scar. 3 - scar contractures across joints result in flexion deformities that cannot be corrected but by surgery. 4 - circumferential scars cause stenosis and non-compliance of tubular structures, in this case of the esophagus after lye ingestion. |
|
|
|
|
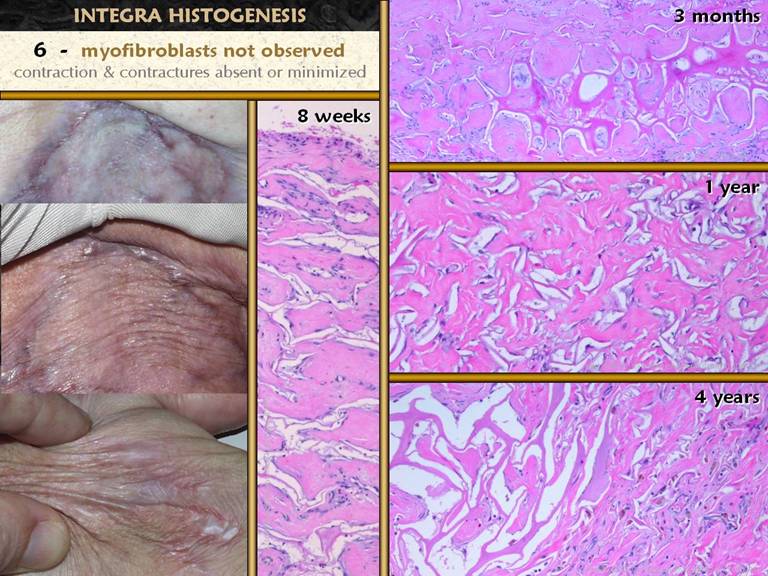
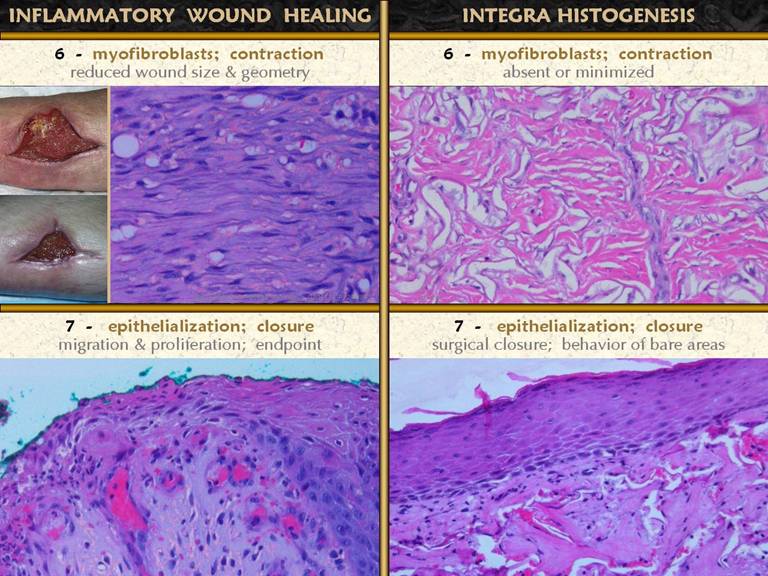
SLIDE 18Myofibroblasts are fibroblast-looking cells which also contain muscle proteins. Their mobility allows them to pull on the wound and contract it. They arise with the other histio-fibroblasts. While they cannot be discriminated with ordinary light microscopy or stains, their effect is clinically very obvious. The photos show an anterior tibial wound which has healed almost entirely by contraction, with only a small area of epithelialization. Refer to the achilles ulcer on the preceding slide. The skin margins are turned inward toward the wound surface, a common finding which is a consequence of contraction. The histology images show the wound margin subjacent to a fold of this kind. Between early wound module at the top (note the streaming vessels) and native fascias below (pink eosinophilic area) is a zone of fibroblast and collagen condensation which is distinct, much denser, straighter, and more lamellar than surrounding areas of fibroplasia. This is the “rubber band” that is contracting the skin above, shown in close up in the middle. |
|
|
|
|
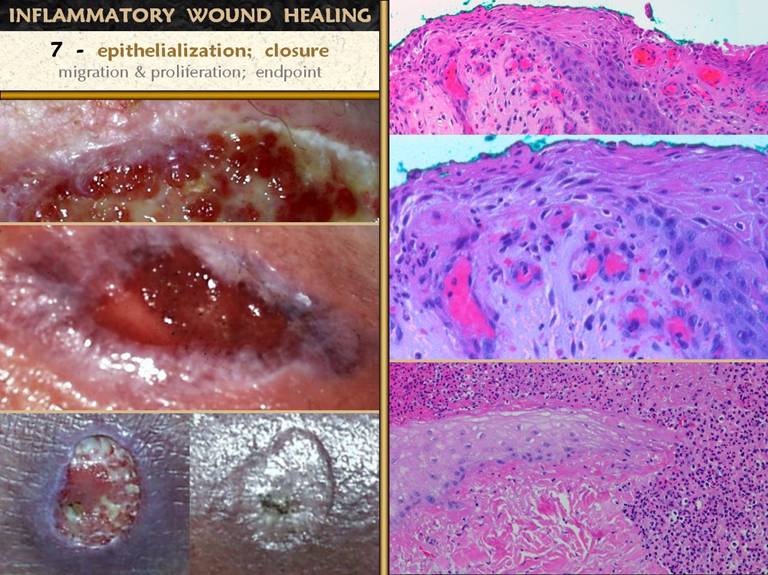
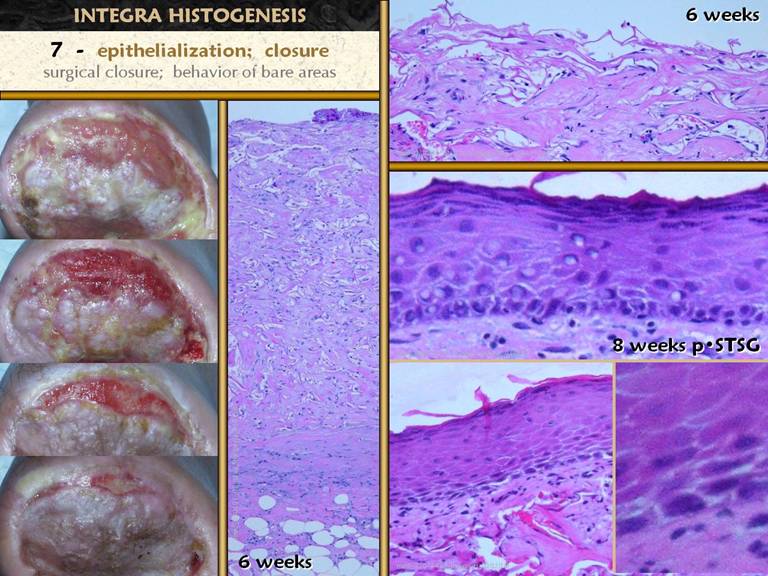
SLIDE 19Closure of the wound means sequestration of the
mesenchymal elements underneath (everything else already discussed, plus all
native fascias) from the ambient world without by a layer of epithelium. Complete epithelialization is the nominal
endpoint of wound healing for the sake of practical everyday wound
management. The photos demonstrate the
process. The upper photo shows that
epithelial outgrowth from surrounding skin edges occurs only where granulation
tissue and other wound module elements have established a suitable foundation
for epithelial cell migration. Robust
active ingrowth is evident in the middle.
At bottom is a small wound that has healed exclusively by
epithelialization rather than contraction - the margins of the ulcerated
dermis are clearly seen, even after it is healed, due to epithelial growth
over the edges and down into the crater. Upper right.
Epidermis at the edge of an open wound. What were normal basal cells and acanthocytes
have become primitive and migratory, streaming outward toward a wound margin
that has a suitable wound module underneath, especially capillaries. Middle right. A
close up view of the above specimen.
Migrating epithelium bears little resemblance to its mature form, but
the cells maintain contact with each other as they spread superficially and
tangentially in an elongated flattened form. Bottom right. Another wound, at the edge of pressure necrosis. The injury is two to three weeks old. This is the edge of the injury. Below and pink is normal living dermis. To the right (and along the top) is a zone of injured but living tissue, filled with acute inflammatory cells. This area will either heal, or separate eschar along the boundary. Above left, dark pink, is dermal necrosis, and eschar cleavage is already occurring at the boundary. Coming in from the left is a spearhead of migrating epidermis. It is growing directly into the damaged interface and is responsible for eschar separation from the margins. The cells are primitive, but maintain a loose basal layer organization, with very thin spindle cells at the leading edge, with rapid turnover and keratin production lifting the eschar above. Numerous mitoses are visible at higher powers. |
|
|
|
|
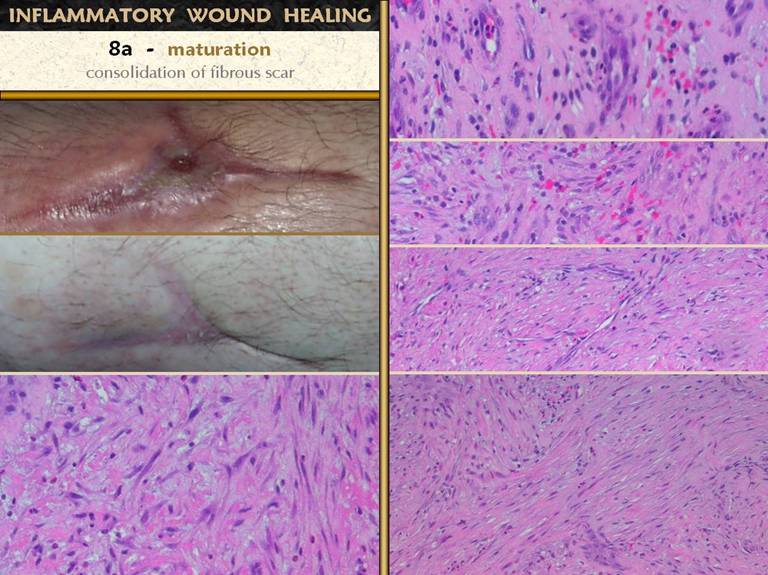
SLIDE 20Once the wound is closed - epithelialized - there is no
longer any source of inflammation or stimulus, and the proliferative phases
of the wound module cease. However,
the various strata of the wound continue going through their programmed
sequences. There are three notable
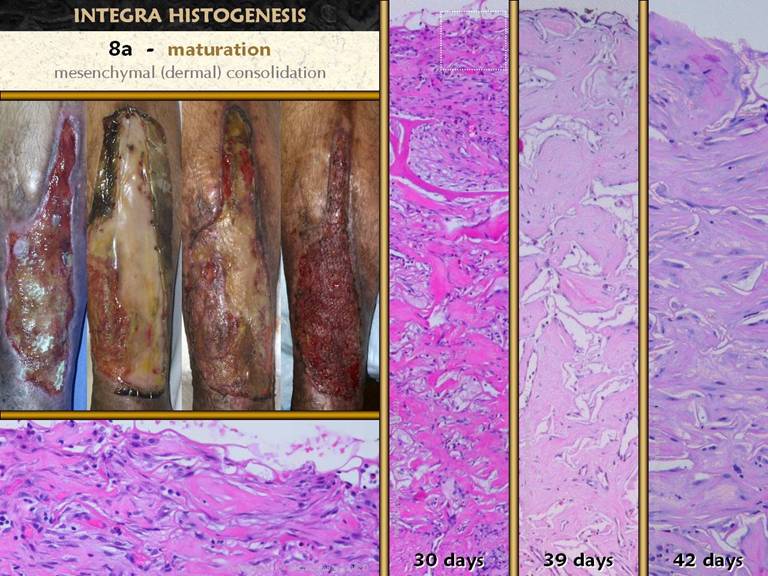
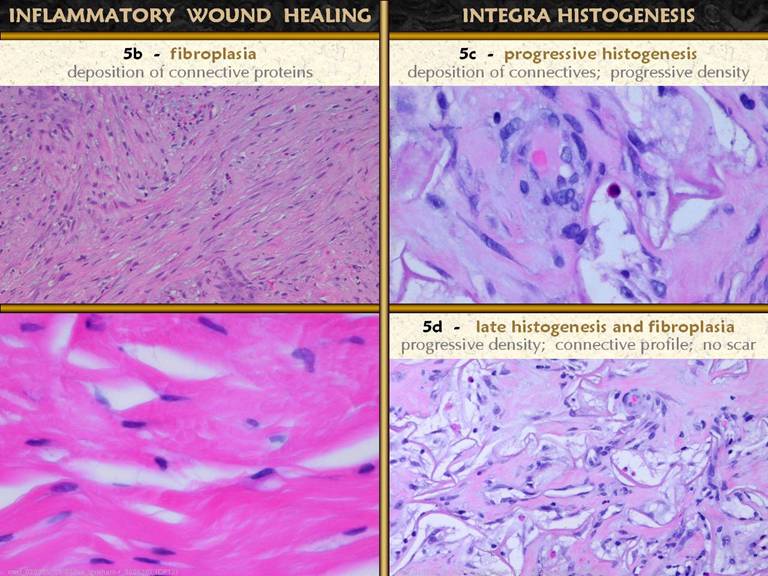
events in the process of wound and scar maturation. The first is the completion of the repair
process leading to consolidation of the fibrosis. The photos show how a young scar can be
observed clinically to contract further and strengthen. On the right is the sequence of fibroplasia
already demonstrated. At top is the appearance of histio-fibroblasts, with early
collagen deposition. Next down shows
an increase in cell and collagen density, with early lamellation and orientation
of the cells and scar bundles. Next
down shows fairly dense cell and collagen packing among very mature
vessels. Finally, at bottom, is the
densest, most non-compliant scar, made from thick, highly stratified
collagen-fibroblast bundles. This is
the peak of the acute scar, having been generated in a time frame of 2 to 4
weeks after initial injury. If there
is no further inflammation or other stimulus to wound module proliferation
(which will continue to make new young scar), this peak proliferative scar
will start to modify back toward something resembling normal dermis or
muscular fascias, a process that will take weeks or months to complete. Bottom left. In all other images, the view is orthogonal to the wound surface - the view is a cross section. The fibrocytes are flattened and layered. They appear spindle shaped in cross-section, but they are actually flattened and wide, evident in a tangential section (parallel to the surface) through a mid zone of the wound. |
|
|
|
|
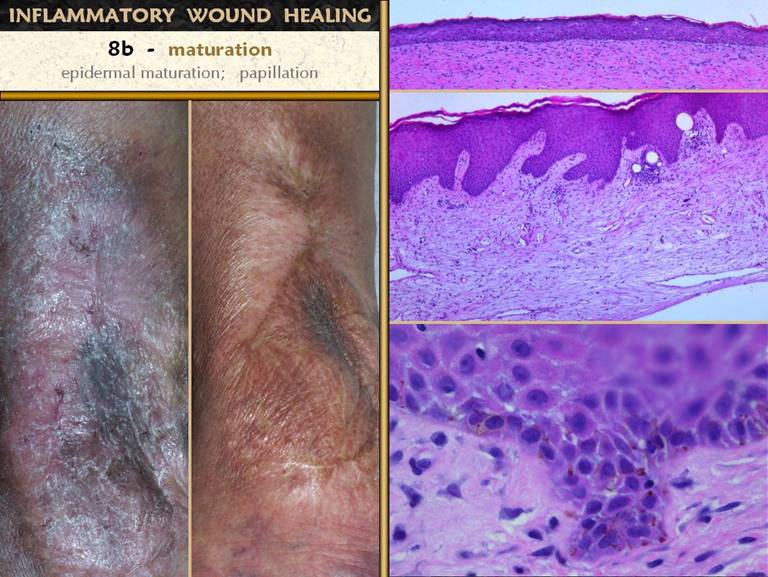
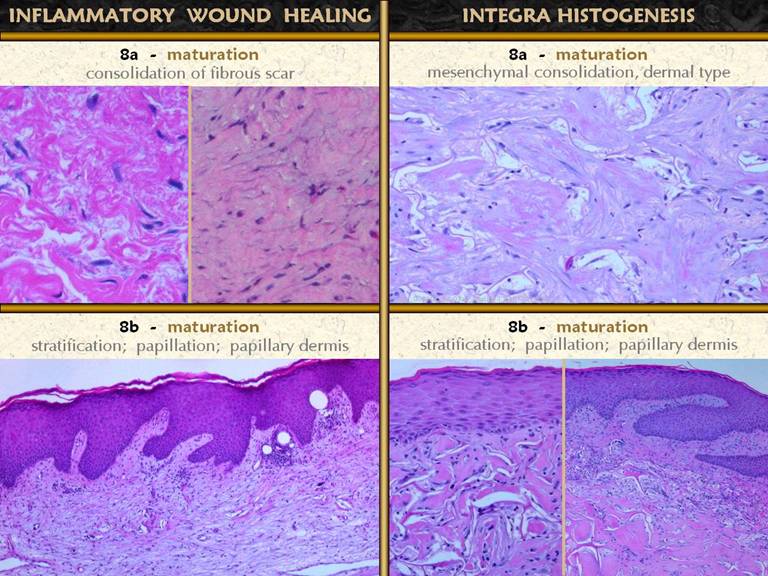
SLIDE 21The second maturation process is the complete
development of the epithelium.
Epithelium appears in two ways - migration from wound margins or by
surgery (skin grafts). Either way, the
young wound has a thin epithelium (epidermis in these images). Once the basal cells reorganize themselves
and resume mature function, several things happen. Acanthocyte proliferation thickens the
epidermis and leads to the formation of rete pegs as vascular tufts tile the
subepithelium to maintain blood supply to the thickened lamina. A specialized lamina of mesenchymal tissue
also develops. In the skin, this is
the papillary dermis. It is distinct
in morphology from the reticular dermis in normal skin. In healing wounds and skin grafts, the
papillary dermis does not appear until epidermis has covered the wound. The papillary dermis is engineered by the
epidermis which acts in effect like the inflammatory and macrophage layers of
the young wound to direct the development of the subjacent tissue. Top right. Young
epidermis after a skin graft. There is
no papillation, and no specific histo-morphology of the subjacent scar. Middle right. A
mature regenerated epidermis. Normal
acanthosis with rete ridges and mild superficial papillomatosis is
present. Blood vessels are present in
each dermal papilla, the vascular tufts which supply the epidermis. The dermal layer has two distinct
tangential zones. The upper layer is
the papillary dermis, triggered by the overlying epidermis when it was placed
on the underlying reticular layer. The
new papillary dermis is fairly normal in appearance - it may improve further
with age, but it already looks like normal native papillary dermis. The bottom reticular layer is NOT at all
like normal reticular dermis. It is
the scar from the previous open wound.
It is cellular and has lamellated collagen which is dense, regular,
and non-compliant, but with relatively thin collagen bundles compared to
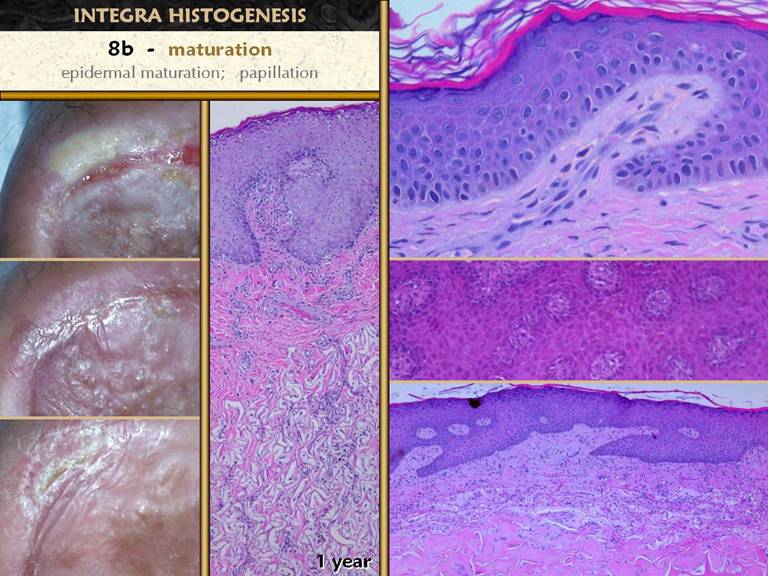
normal reticular dermis - i.e. it is scar. Bottom right. As
epidermis matures, other normal features appear, such as Langerhans cells
and, depending on the source of the new epithelium, melanocytes and
melanin. These are all functions of
the epidermis and normal epidermal-dermal interactions, and they occur independent of what had previously happened in the
mesenchymal dermis or scar or wound module underneath. The photos show an ankle ulcer closed with a skin graft. It’s appearance one year later shows how the epidermis matures, corresponding to the changes seen in the histology views. |
|
|
|
|
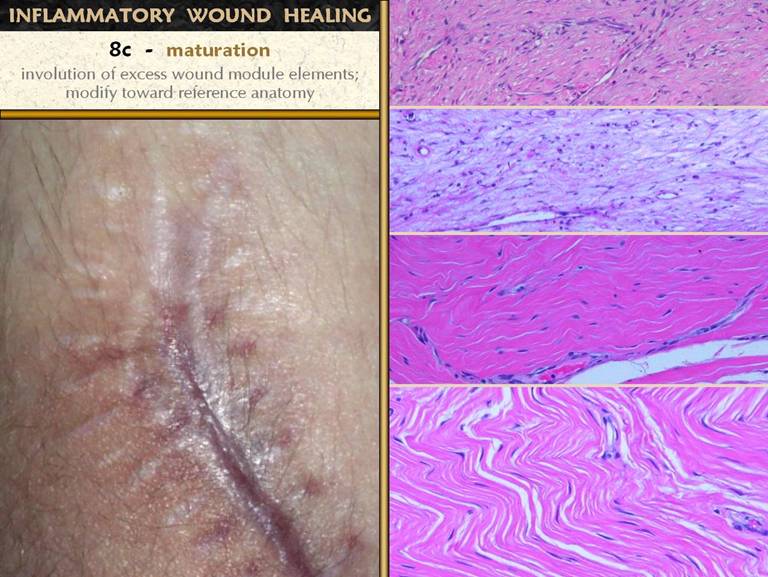
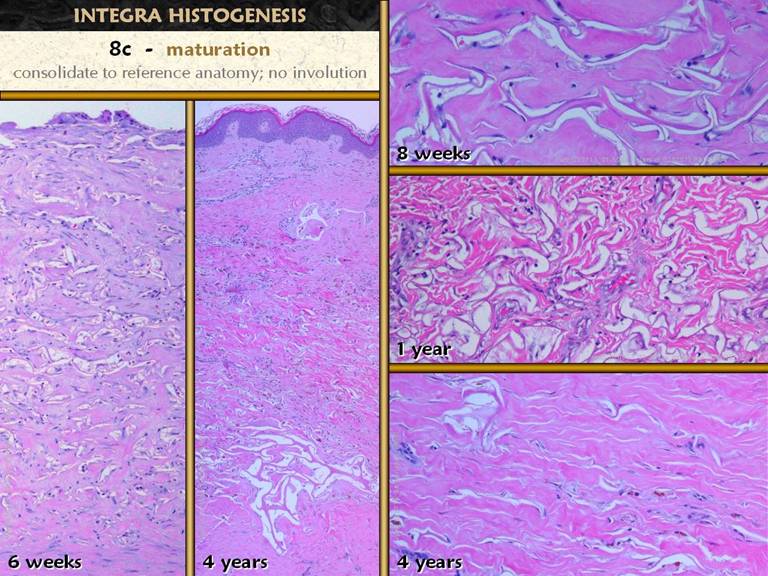
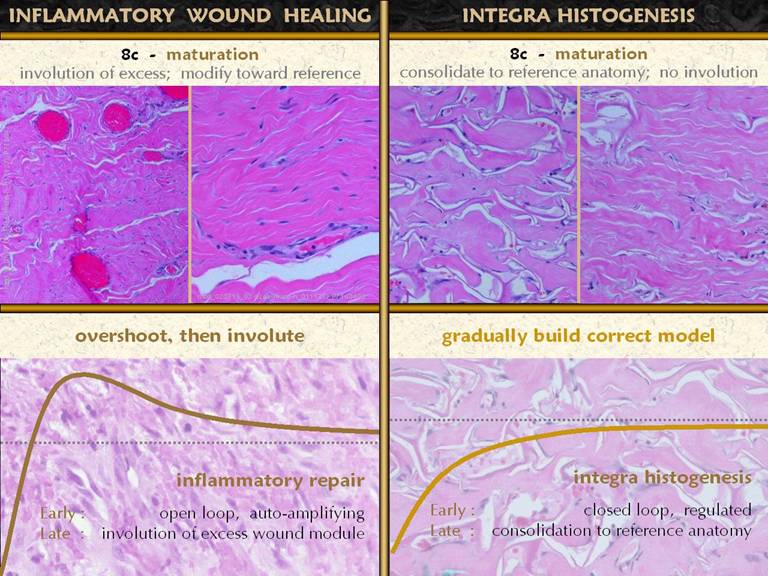
SLIDE 22The final maturation event is that which is usually meant
when when talking about scar maturation - the long term involution of the scar. The early healed wound has all of the
collagen, fibroblasts, and excessive blood vessels seen in all of the
previous images. All of these elements
are over abundant compared to any normal tissue. As the healed wound ages, the excess
materials are removed, and gradually the scar takes on characteristics closer
to normal skin and fascias. The photo
shows a set of scars from an area having had multiple operations. Some of the scars are young, and some are
old and mature. The older more mature
ones are pale and flat, soft and compliant.
The younger ones are thick, stiff, and discolored from vascular
plethora. Right top. Fibroblasts,
collagen, and new blood vessels at the peak of proliferative repair. Right second. The
“reticular layer” of skin scar after it is fully epithelialized and the
epidermis itself is healthy (same specimen as on preceding slide). Vascular density seems to be less, and
cellularity in the collagen also seems less, compared to their peak density
in the top image. Right third. As a
scar becomes fully matured, collagen bundles become wavy and springy, with
tangential spaces or planes between bundles.
Vessel morphology is very mature, and the number of vessels is
diminished back to a normal vascular density, meaning that clinically the red
color has faded. Fibrocyte density is
much decreased. Right bottom. In the fully matured scar, herringbone patterns attest to a final collagen configuration that is once again compliant and mobile. Vessels are sparse, and fibrocyte density is at a minimum. While not looking exactly like normal dermis or musculotendinous fascias, it looks very similar. |
|
|
|
|
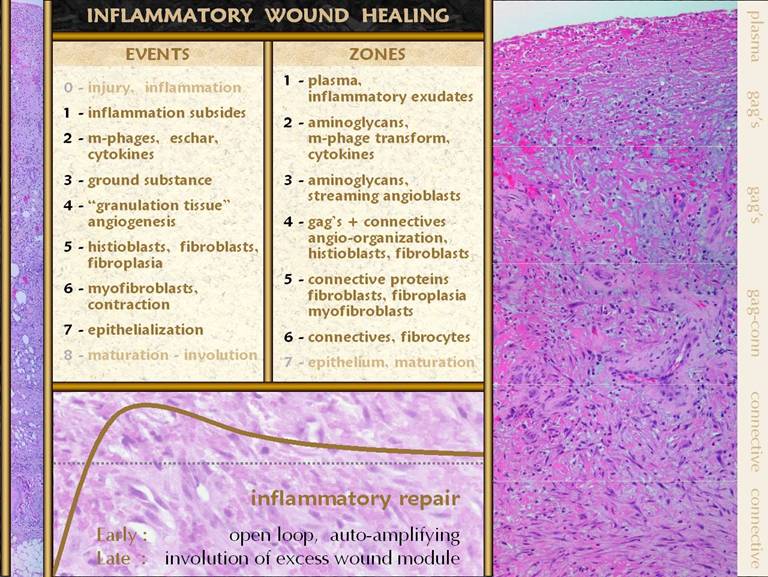
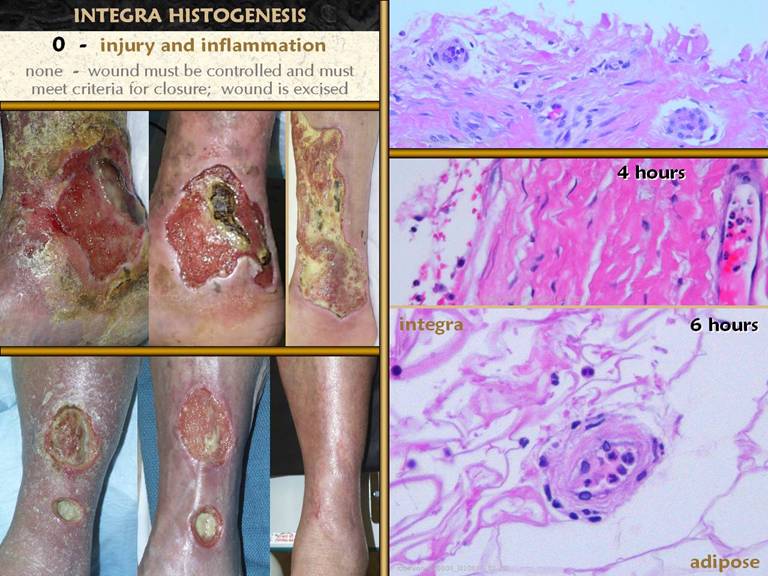
SLIDE 23SUMMARY of Normal Inflammatory Wound Repair Injury triggers inflammation which begets the repair
process. It is an orchestrated process
referred to as the wound module, and the significant events are: 0 - injury and inflammation trigger the process. 1 - inflammation subsides. 2 - monocytes transform to
macrophages which have two jobs, the first being to phagocytize and separate
any eschar, and the second being the production of cell stimulating cytokines
to activate local histoprogenitor cells.. 3 - ground substance substance
appears so that recruited cells have an environment in which they can
function. 4 - angiogenesis begins as
macrophage cytokines stimulate nearby angiocytes. Angioblasts stream toward the macrophages
and then reorganize into blood vessels, creating an environment in which
other histioblasts can then perform their functions. 5 - the angiopericytes also give
rise to the histioblasts, which once in the wound, coming in behind
established vessels, begin to mature into fibroblasts which make the
connective proteins which restore mechanical stability and integrity to the
wound. 6 - specialized myofibroblasts
also arise, causing the wound to contract. 7 - epithelial proliferation and migration occurs on the
surface of other established wound module elements, eventually closing the
wound, 8 - once the wound is
epithelialized, the wound matures, first as the continuing consolidation of
the scar and maturation of the epithelium, followed by involution of
excessive cells and proteins deposited during the proliferative repair phase. These events can all be observed histologically, and they
occur in several distinctive zones or strata within the wound. Remember that in a normal healing wound,
depth equals history, and therefore a vertical slice of the wound represents
the entire repair process in sequence.
The recognizable strata are 1 - the top or surface layer, a coagulum of fluids and
plasma proteins, populated exclusively by acute inflammatory cells, 2 - a transformation zone where monocytes are converting
to macrophages, aminoglycan ground substance replaces the plasma coagulum as
the ambient medium, and the new macrophages start to make chemotactic
cytokines, 3 - a zone of streaming angioblasts, arising from
subjacent blood vessels, and migrating up through the aminoglycan ground
substance toward the source of cytokines above, 4 - a zone of angio-organization, where re-established
blood supply makes a haven for young histioblasts to proliferate and begin
the transformation to fibroblasts, where thin collagen begins to replace
ground substance, 5 - a zone of fibrous proliferation, where fibroblasts
become abundant and start to make dense connective proteins, and where wound
contraction can occur due to the effects of myo-(muscle proteinated)-fibroblasts, 6 - the fully developed scar, where fibroblasts become
mature fibrocytes, and collagen is dense and takes on a stratified
architecture, 7 - epithelium grows on the
surface of this wound module, from the margins of surrounding skin, and as
the epithelium closes, the wound begins attritional maturation throughout. Inflammation and inflammatory wound repair are a
coordinated response to injury that starts with a big bang. The onset and development of inflammation
is an auto-amplifying process that dumps huge numbers of cells and
pro-inflammatory chemicals into the wound in a very short time. The reparative process is likewise
characterized by aggressive, rapid, highly cellular proliferation of
stimulated cells. In a healthy acute
wound in an unimpaired host, monocyte-macrophage transformation (stratum 2)
is in progress by three to four days after injury, angioblasts and early
angiogenesis (stratum 3) can be seen grossly by 5-6 days, clinical signs of
wound adhesion due to connective proteins is evident at 7-10 days (stratum
4), a wound able to withstand ordinary daily loads without sutures is present
at 10-15 days, and a stable scar with dense collagen is present in 15-20
days. Peak consolidation of the scar
is evident at 4-8 weeks, and involution and maturational remodeling proceed
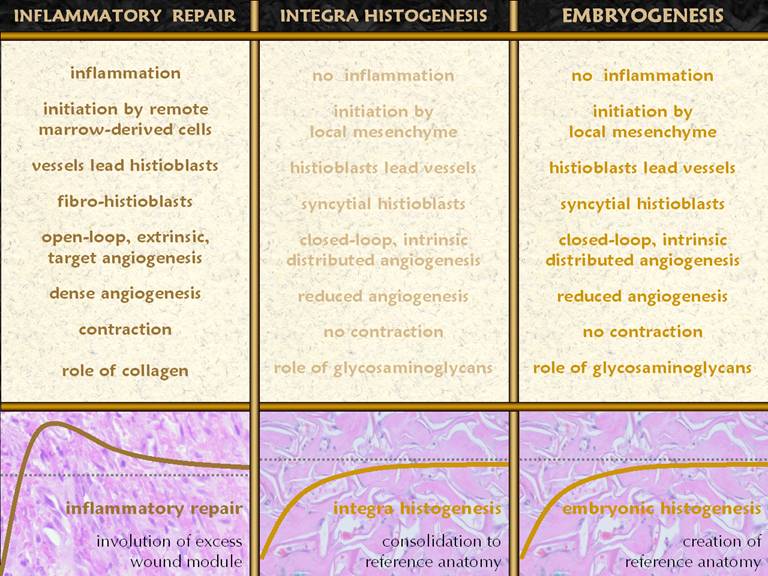
from there. Bottom graph: This
shows the condition of the wound, some vague indistinct measure of quality
and quantity, versus time after injury.
The dotted line is a target level representing the quality and characteristics
of normal skin. The graph shows the
behavior of the repair process, beginning at the beginning with not much
“stuff”. What the inflammatory wound
does is to go overboard, depositing large amounts of cells, vessels, and
connective materials, rapidly building a dense scar which binds the wound
together, but with unfavorable characteristics which are unlike normal skin
and fascia. Only after the scar is
stable and closed does the host modify the scar, slowly returning it toward
something more like normal fascias. These concepts and observations will be compared,
head-to-head, one-by-one, with repair and regeneration in Integra. |
PART 2: INTEGRA HISTOGENESIS
|
|
|
|
|
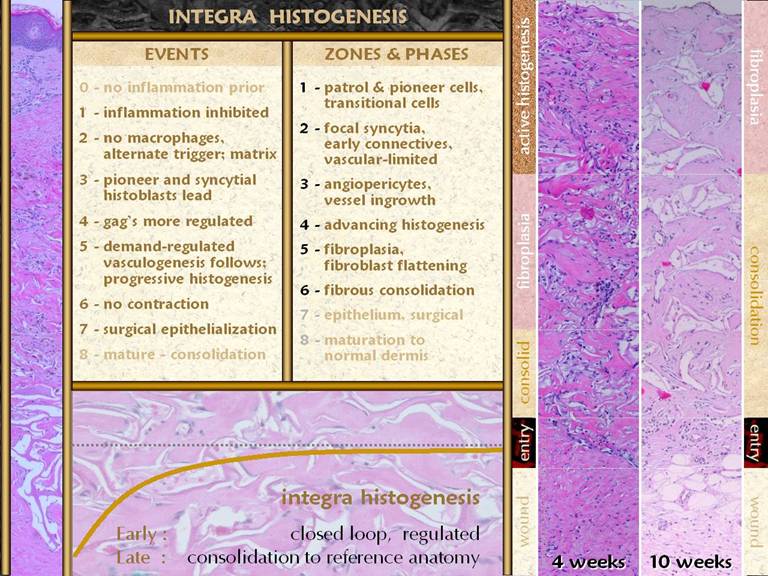
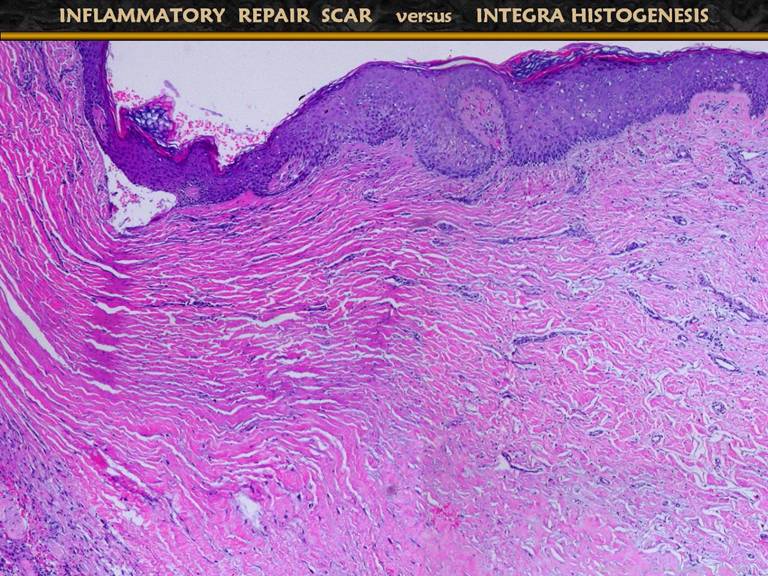
SLIDE 24The photos show a muscle flap and skin graft over a ruptured achilles tendon. The graft is hypertrophic and ulcerated, typical of any scar across a flexion surface. The micro image shows what regenerated Integra looks like histologically. The nice result after Integra reflects that regeneration of this material is inherently different than normal scar or wound repair. The case will be made that the regeneration of Integra is a process analogous to embryonic histogenesis. Using the sequence and events of inflammatory wound repair as a basis of comparison, the events of Integra histogenesis will now be examined. |
|
|
|
|
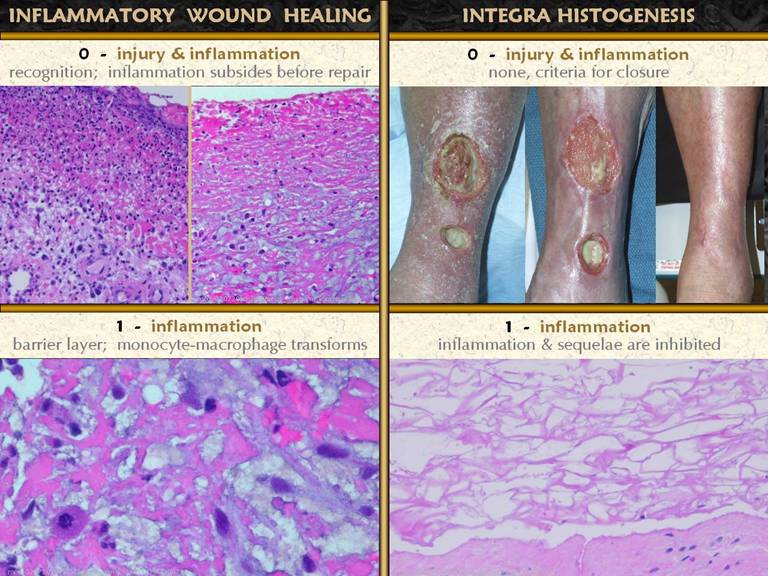
SLIDE 25Injury, disease, and inflammation must be controlled prior
to any attempted wound closure, and that is as true or more so for
Integra. The notion that injury
triggers inflammation triggers repair is irrelevant to using Integra, because
injury and inflammation are controlled a priori, and neither directly induces
a “state of integra”, only the surgeon does. Photo panel, top left and middle. An example of a chronic venous wound
carrying a recent deceased skin graft.
Attempts to graft the wound as it was was
foolish. After 2 weeks of good care,
dermatitis, wound exudates, and edema are all controlled. The patient was then eligible for surgery,
in this case, excision and successful skin reconstruction with Integra. Photo panel, bottom.
This patient did not have Integra, but it illustrates the point which
is so essential to good wound care and wound surgery, including Integra. This healthy patient had a minor leg
laceration, treated aggressively but ineptly with noxious topical agents,
leading to severe dermatitis, wound perforation into the anterior
compartment, and compartmental abscess which then drained itself inferiorly
over the tendons. The patient was two days away from leg amputation, to be performed
by the same bozo who messed up the sure-to-heal boo-boo in the first
place. Two weeks of good skin and
wound care restores everything to health, and then the tendons were closed
with a minor local flap - healed. The
importance of proper skin and wound care and good preparation prior to any
type of wound closure - either surgical, by natural contraction, or with
pharmacological support - cannot be overemphasized. As a foreign or semibiological material
which is not alive and cannot intrinsically heal itself, and which is also
expensive, it cannot be abused, wasted, and lost by inadequate preliminary
care. Central photo and top right micro image. All Integra usage should ideally look like
this. The leg and ulcer, due to
rheumatoid, are clean, proliferating normal wound
module, and the periwound is free of gross inflammation and edema. It took several weeks of care to get to
this point, where the wound can now be excised and skin reconstructed with
Integra. The micro picture shows the
wound surface after excision, the surface that the Integra went onto. There is divided collagen, normal
fibrocytes, and normal blood vessels, but not a single inflammatory
cell. This wound is ready for closure,
with Integra or by any means. Right middle. This
patient had dermatofasciectomy of both lower extremities for lymphedema. The biopsy was taken form the surface of
the first extremity, just prior to Integra placement, after an interval of
several hours during which the second leg was excised. The view is of normal sural fascia. Neutrophils are marginated and static in
the adjacent blood vessel, and neutrophils are already present on the wound
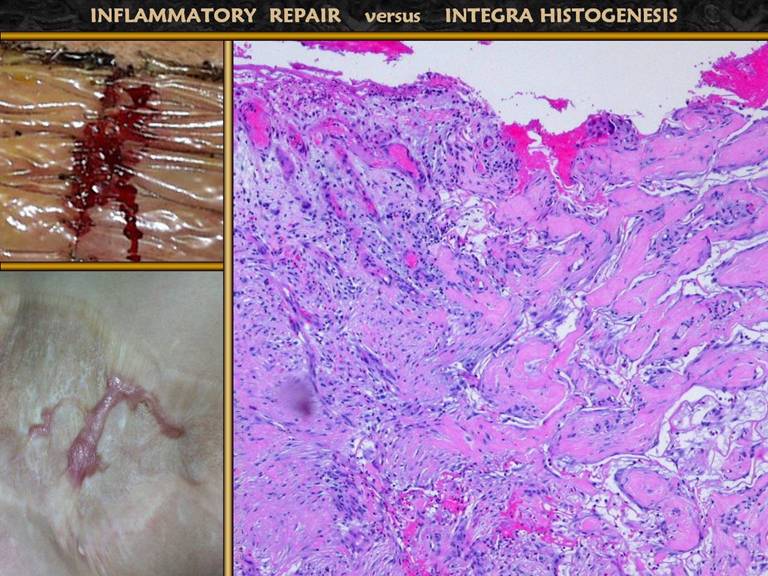
surface. Right bottom. The same patient, two hours later, after the legs are cloaked in Integra and wrapped in compression bandages. The biopsy was taken at the end of surgery, through a small window in the dressings. The image shows the Integra-adipose interface, with a vessel at the surface. Marginated neutrophils escaping into the Integra are abundant. There was an injury - the surgical excision - and the wound is clearly aware of the injury, because inflammation has begun. If this was a normal wound, it would progress to all stages of the normal inflammatory-proliferative wound module. One might guess that the inflammation will become more intense, but this is Integra’s first bit of magic . . . |
|
|
|
|
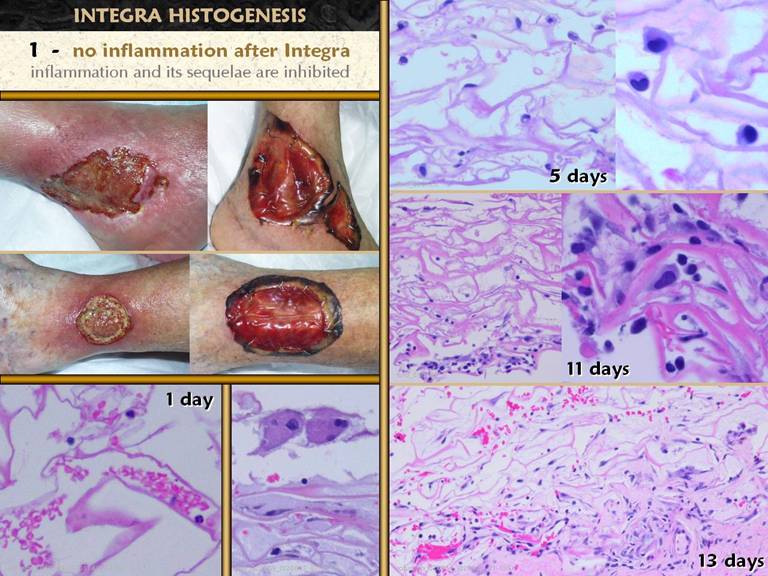
SLIDE 26 . . . while the inflammation might have been triggered due to
the injury, Integra has the property of turning inflammation off. The two patient sets show chronic ulcers at the peak of
their preparedness just prior to and a week after wound excision and
Integra. These are not ideal wounds,
but it was as good as it could get for these two, and is one of the reasons to
use Integra - its ability to control inflammation and pathergy. Note that refractory inflammation is
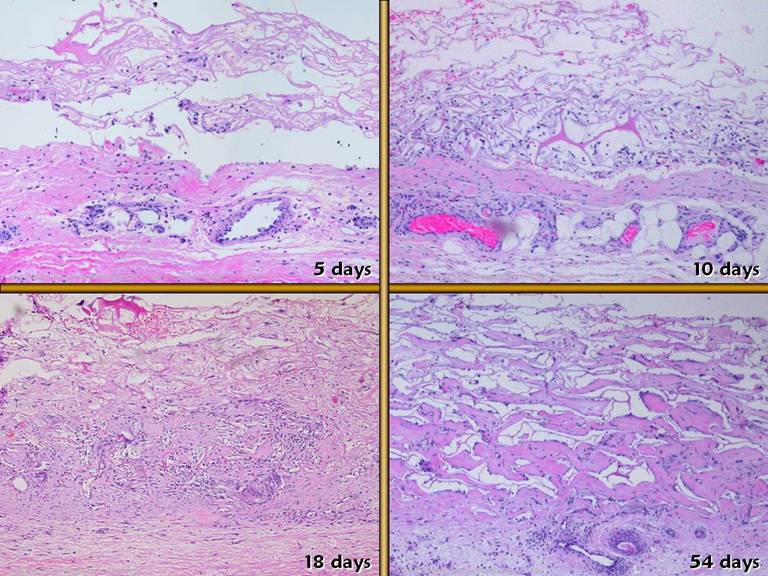
completely resolved with Integra in place. Bottom left. A
biopsy at one day shows a few red and white cells in the matrix, due to blood
absorption into the sponge during surgery.
Leukocytes are no more prevalent than expected for its normal numbers
in whole blood. There are no signs of
inflammation in the matrix. Upper right. Within
4-5 days, the matrix is peppered with small round cells. While they somewhat resemble lymphocytes,
there is simply no evidence of inflammatory type leukocyte infiltration, and
there are clearly no neutrophils, as one would anticipate with any acute
inflammation. In the right middle and
lower panels, later phases of regeneration are in process, but never (unless
there is some complication), do acute inflammatory cells appear. Lower middle. These
are the start of foreign body giant cells, common at the top of the matrix
adjacent to the silicone, and seen within the first few days, until the
silicone is removed. One of Integra’s important properties is that it suddenly “turns wounds off”, immediately making the injury “not a wound” in the sense that the integrated response to injury, inflammation and fibrous repair, immediately ceases and does not occur. When patients immediately feel good and have surcease of pain, when pathergy, inflammation, and tissue necrosis and lysis are immediately arrested, when the wound immediately reorganizes into a process comparable to embryonic histogenesis and the normal wound module is suspended, these are all important events indicating that the programmed response to injury is simply turned off. The injury is still an injury, and it may still be a wound in the sense of injury or disruption of mechanical integrity, but in terms of its physiological response and behavior, a wound under Integra ceases to be a wound. |
|
|
|
|
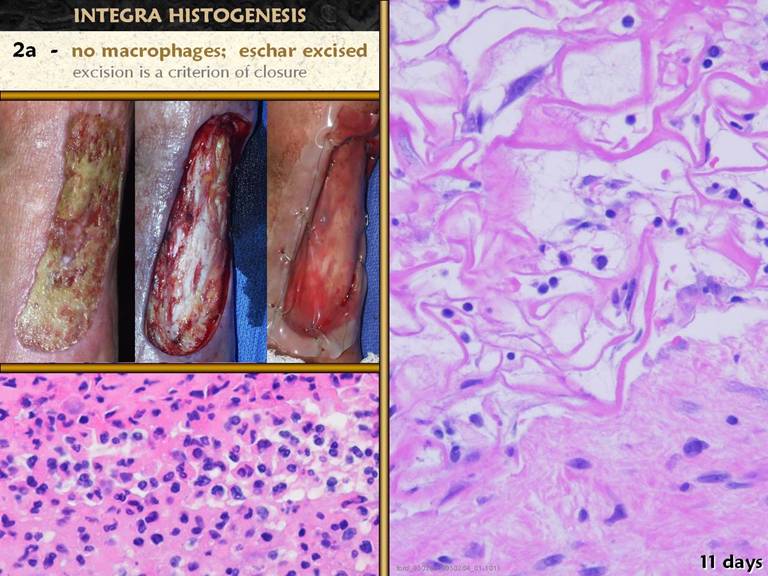
SLIDE 27Lower left. Recall how crucial monocyte-macrophages are to inflammatory wound repair, shown here in their afferent role as a wall of phagocytes separating eschar. With Integra, eschar is an irrelevant concept, because it is completely removed prior to placement of the material, as shown in the photos. Because there is no blood-borne inflammatory process, monocytes and macrophages do not appear. The right panel shows healthy early Integra regeneration, at the interface with the host tissue. It is a bland interface - there are no phagocytic cells nor any other acute phase cell accumulating in, at, on, or otherwise reacting to the material. |
|
|
|
|
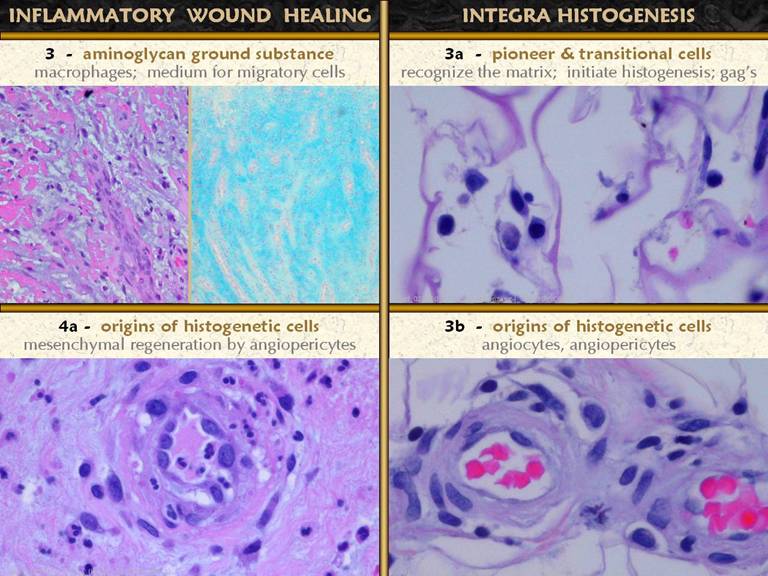
SLIDE 28Regeneration of Integra obviously depends on some initial
recognition of its presence and properties.
If inflammation is not present, then inflammatory mediators, including
platelets, leukocytes, and acute phase protein cascades, cannot be the agents
of recognition nor the triggers to regeneration. What then does recognize the material and
initiate regeneration? Lower left shows the early regenerating matrix, subjacent
to which are blood vessels. Angiocytes
and angiopericytes have undergone a massive hypertrophy, and are the source of the histogenetic cells populating the
matrix. The lower right shows empty
matrix and early transmigration of cells into the matrix. The upper right shows the types of early
cells which populate the matrix:
pioneer and transitional cells which become histioblasts, and
angioblasts. What turns this system
on? How do the emissary cells know
what to do? The current study does not have sufficient specimens from
the first few days, nor special stains which might
identify the first cells to recognize the matrix. Because the system is devoid of acute
inflammation, and because the matrix is insoluble and non-diffusible, it is
hypothesized that there are “patrol cells” that normally wander the soft tissues, and that upon randomly finding the matrix, they
initiate a response. What tells these
or whatever cells to respond without inflammation, and to initiate
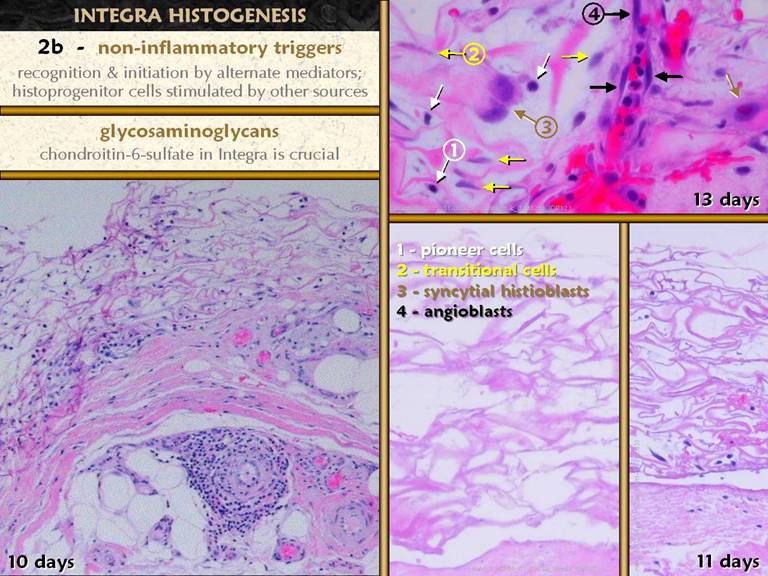
histogenesis? Collagen in Integra provides structural stability, but none of the very many other collagen products that are used in surgery have the ability to transform repair the way Integra does. Because of the known central role of aminoglycans in embryogenesis and fetal wound repair, it is hypothesized that the chondroitin-6-sulfate in the matrix is the crucial flag which tells the hypothetical patrol cells to initiate the histogenerative sequence. |
|
|
|
|
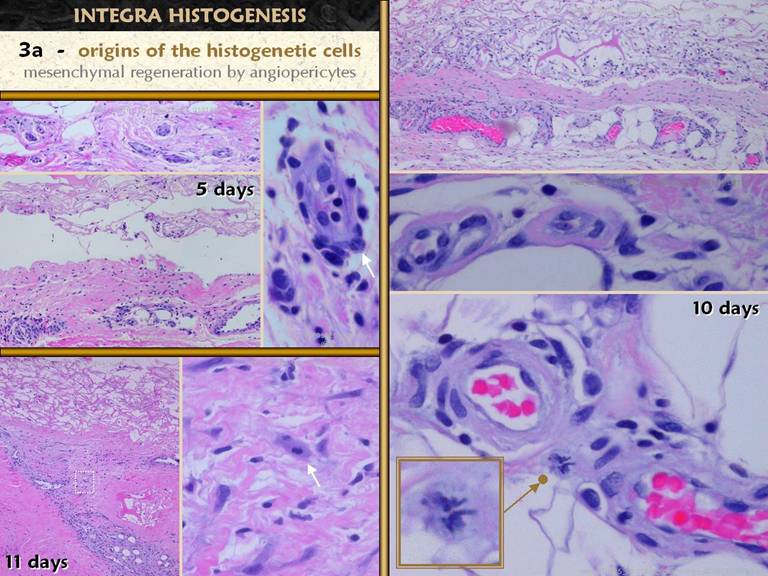
SLIDE 29Concerning the origin of the histogenetic cells, this is a
closer view of how they arise from angiocytes. Above left, normal vessels in normal fascia at the time of
wound excision, just prior to placing Integra. Left middle, at five days, the perivascular
zone has proliferating and hypertrophic angiopericytes, with cells emanating
from there, migrating into the matrix.
The close up shows how these cells are responding, with increased cell
size, increased nuclear size, stippled chromatin, and even hypertrophy of the
endothelial cells. Lower left, another
view of massive angiopericyte hypertrophy, with a dense stream of cells going
to the matrix; the
zoom in view to the right shows how these stimulated cells are both migrating
and dividing, evidenced by a mitosis. Right lower, another view of angiopericyte hypertrophy, including another beautiful mitosis. Right middle shows endothelial hypertrophy. Right upper shows that histogenesis occurs in a patchy distribution, being densest at first wherever there are subjacent vessels and angiohypertrophy that can source cells to the matrix. Streams of entrained cells can be seen going from vessels to matrix. |
|
|
|
|
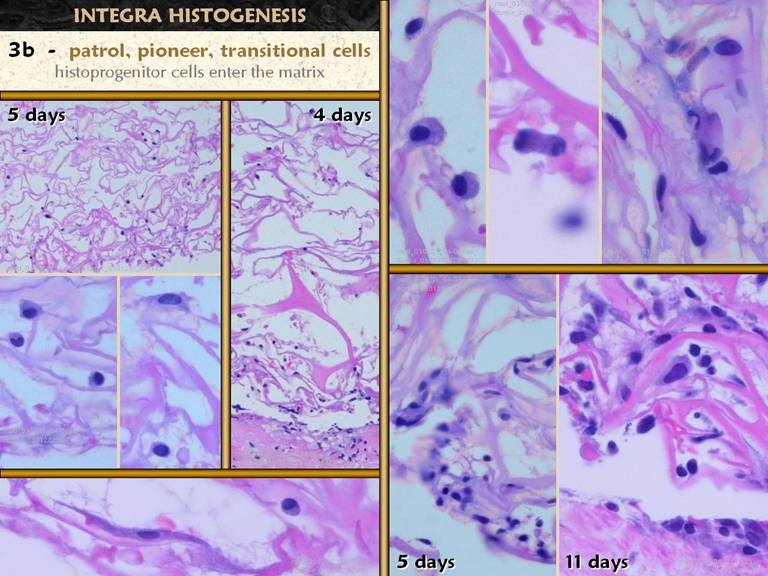
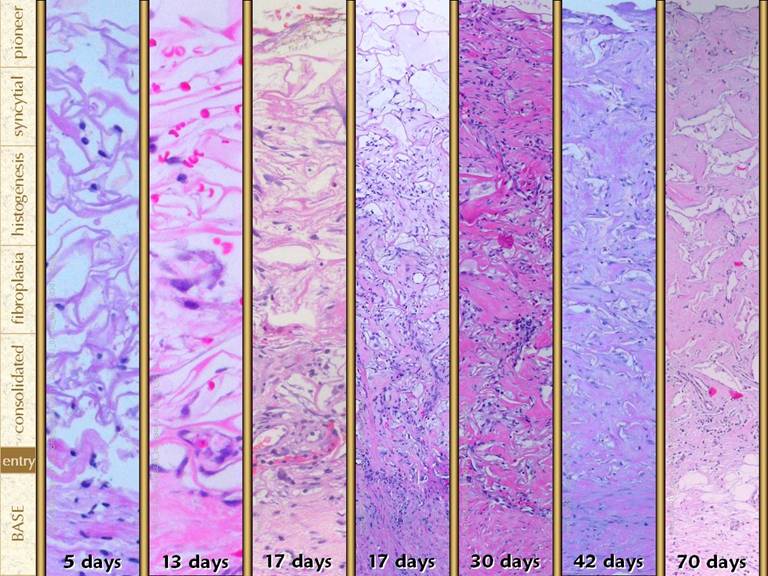
SLIDE 30Regardless what recognizes the matrix and triggers the response, once the response is initiated, the process is is easy to track histologically. It begins with small round cells which pepper the matrix. These early “pioneer cells” may have been the hypothetical patrol cells, or they may be angiopericytes mustered by the earliest ones, or both. The left side and upper right panes illustrate these small cells with limited cytoplasm. Note that these cells are evenly distributed throughout the matrix, vertically, from wound side to silicone side. The next phase is for these cells to adhere to the matrix, recognized by their flattening and elongating (left center, left bottom, and others). The upper right pane shows an accumulation of these “transitional cells”. The two lower right panes show increasing cell density. Some of these accumulating cells are new recruits of cells emanating from vessels in the wound, and some are mitotic daughters of cells already present. The upper middle right pane shows one such cell captured during the evanescent telophase. |
|
|
|
|
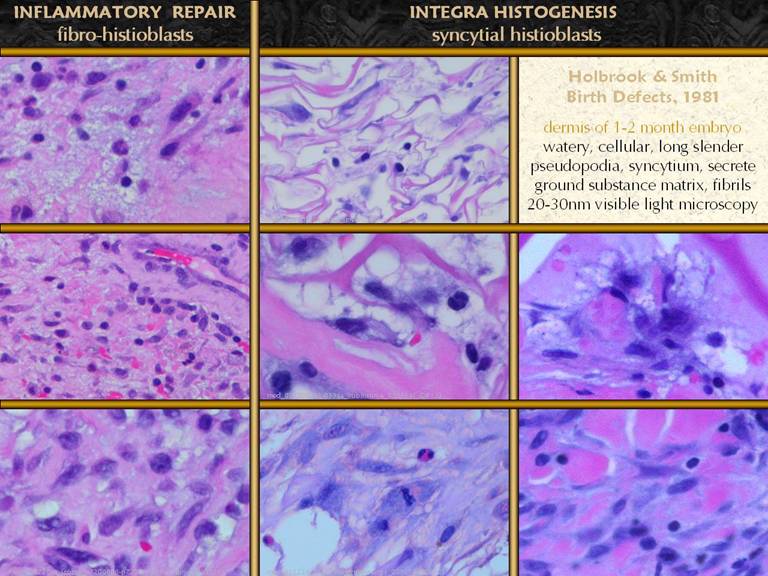
SLIDE 31The small meandering pioneer cells finally adhere to the
matrix and undergo a transitional phase. What are they transitioning to? They begin to accumulate cytoplasm and
nucleoplasm, becoming very large, in preparation for proteogenesis. They have long interconnecting pseudopodia,
and seem to have indistinct borders as they group themselves together. While they do not form a true syncytium (cf
foreign body giant cells), they appear to so (as described in embryonic
studies literature), so the term will be used here. These new enlarged cells are the “syncytial
histioblasts”. They begin the real
work of making new tissue. In these images, they are the big, pale irregular
cell. Early ones may be solitary, but
clusters are the eventually the rule.
These cells make aminoglycan ground substance (the pale purple
acellular masses along the matrix on the right panes), and they make
fibrillar collagen (pale pink staining amongst these cells. Right mid and lower.
These syncytial cells occupy sponge domain, making early unorganized
collagen. The zoom in view (lower right) shows collagen loci wrapped around
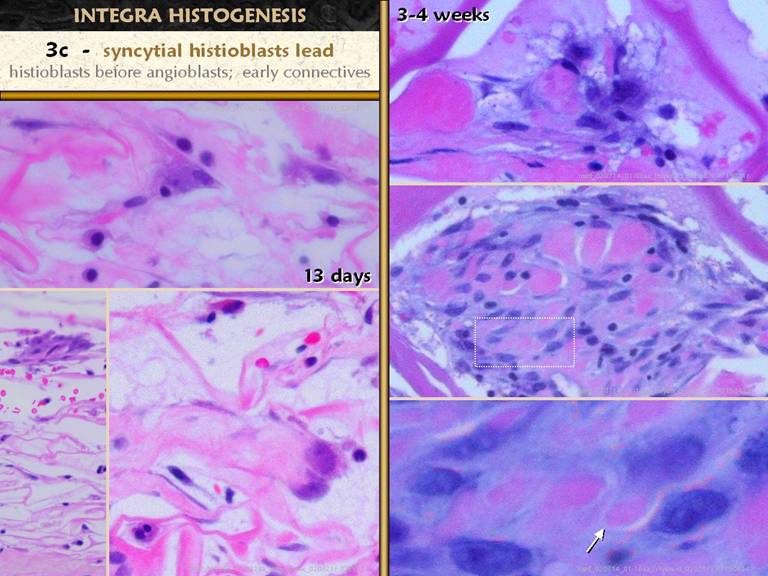
the pseudopods that are secreting them. Lower left. A cluster of syncytial histioblasts high in the matrix, away from the wound base. These cells proliferate and cluster until their mass or the mass of nearby clusters consumes oxygen and substrate to the point that further growth cannot occur until angiogenesis and vascularization occur. |
|
|
|
|
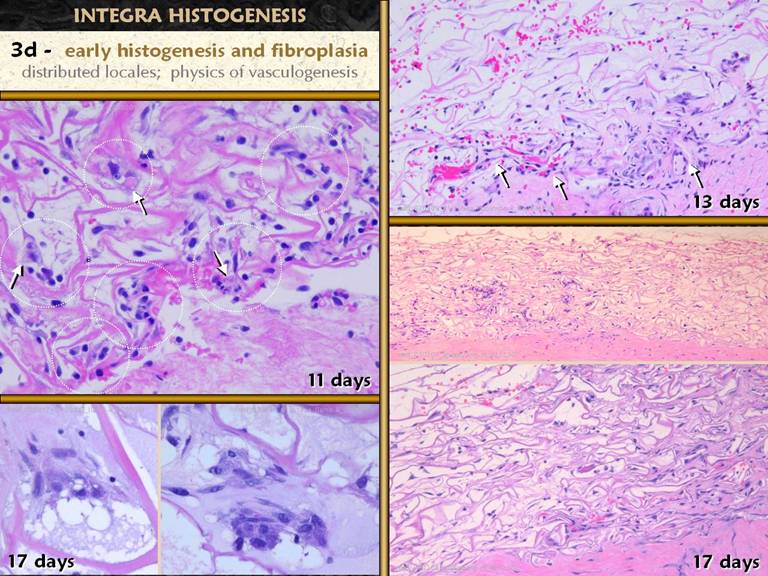
SLIDE 32These images further demonstrate that the early phases of
histogenesis are limited by lack of supporting blood vessels. The early clusters of syncytial
histioblasts are distributed here and there through the matrix, with lots of
empty space between. Left lower shows
typical clusters at the limits of what they can sustain with vascular
supply. Left upper zooms out to show
numerous clusters, marked by dotted circles.
So far, there is no bulk collagen, no filling of the sponge, and no
vascular ingrowth from the host. This is the phase of early histogenesis, characterized by the syncytial clusters making some ground substance and fibrillar collagen. The physics of vasculogenesis dictate that revascularization must now occur, triggered by these cell clusters making diffusible angiogenic factors which are chemotactic to angioblasts. Progressive mass-accumulating space-filling histogenesis occurs only after vasculogenic new vessels reach the clusters. Right upper shows vascular ingrowth into a locale, and space-filling collagenization can be seen in the surrounding zone. Right middle shows the same thing, zoomed out, so that several such locales can be seen. Right lower shows what comes next, the coalescence of these locales, and progressive histogenesis through the matrix. |
|
|
|
|
SLIDE 33In a normal wound, glycosaminoglycan ground substance must
appear early, because in the absence of a connective protein matrix, this is
the medium which early histogenetic cells to create a suitable environment
for their own proliferation, migration, and functional activity. In the early wound, aminoglycans are
abundant until progressive cell proliferation and fibroplasia fill up space. Mature tissues and even scar have some
residual aminoglycan as part of the interstitial “ground substance” between
cells and connective protein fibers. Left upper shows regenerated Integra with the silicone
peeled off, at the time of skin grafting.
The soft moist surface and scattered light reflex is comparable to
that seen on top of a normal wound, which is characteristic of the upper
aminoglycan layer, but unlike a normal wound, mucoid exudates and mucus
stringers never appear. Left lower. The
early cells in the matrix, pioneer and transitional cells seem to float in
the interstices. There is no staining
to suggest the presence of proteins nor
aminoglycans. Presumably the matrix is
filled at this point with serum-like interstitial fluid which fills the
matrix from the underlying tissues by simple hydraulic conductance. Because there is no inflammation, a
plasma-like proteinaceous exudate cannot be present, and there are no other
antecedent cells which could make a gag ground substance for these early
cells to migrate in. Also, recall that
with normal inflammatory wound repair, there is a “lag phase” of 3 to 5 days
between injury and the proliferation of wound module elements, the time
required for inflammation to subside and macrophages to appear to begin the
repair process. In Integra, the early
cells appear during what would be the lag phase in a normal wound, in advance
of when the aminoglycan layer would form. Center. By 11 days
after injury, a normal wound would have a fully developed aminoglycan layer,
yet in the Integra matrix, there are still no signs of any type of material
infiltration of the matrix. In
Integra, the Integra sponge IS the matrix that progenitor cells need. In this image, the cells are starting to
become syncytial histioblasts, and these cells will make the aminoglycans
that are needed for the interstitial matrix between developing fibroblasts
and connective proteins. Right. Between the matrix itself and the nested clusters of cells and collagen, there is a zone of pale lavender-colored material which has enough form and solidity, to have conformed to the matrix (the light blue gaps are a fixation artifact as the material separates from the matrix during processing). This formed substance is presumably an aminoglycan ground substance.
|
|
|
|
|
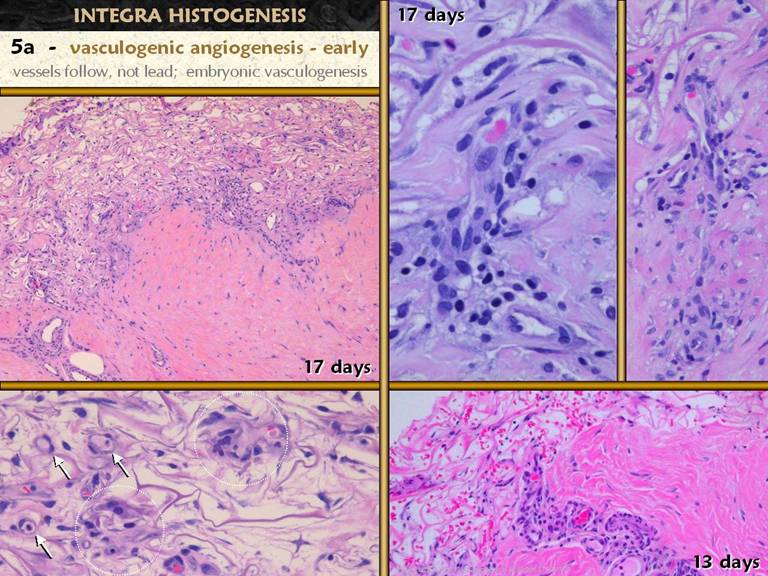
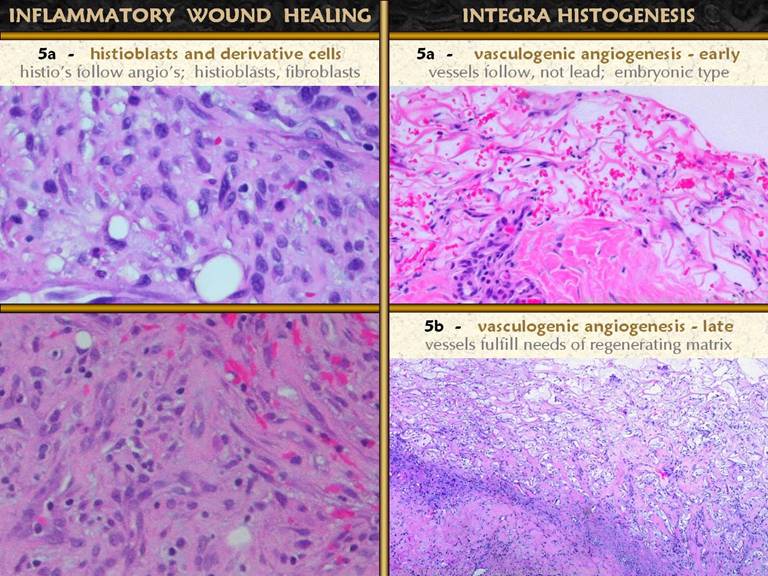
SLIDE 34During normal wound repair, angioblasts and vessels are
the first proliferative local histogenetic cells to appear in the wound. Fibro-histioblasts appear after that. In Integra, non-vascular histogenetic cells
appear first, the syncytial histioblasts.
These cells proliferate into small clusters which, consistent with the
normal physics of cell proliferation and vasculogenesis, can become only so
large until new blood supply is attracted.
This is how blood vessels and the vasculature develop during normal
embryogenesis, and it is what happens next during Integra histogenesis. The clusters of histogenetic cells begin to
make angiogenic cytokines, and the nearest surrounding vessels will respond
to this chemotactic stimulation by sprouting angioblasts and new vessels,
which grow tropically toward the source of stimulation. Left lower. Two
early histogenetic clusters are circled.
The rest of the matrix does not yet have significant cellularity,
collagen, or histio-density, but small reorganized capillaries can be seen
adjacent to the clusters (arrows). Right upper and middle.
Two examples of vessels in the wound base sprouting branches up into
the Integra. Hypertrophy,
proliferation, and migration of angiocytes is
present. Right lower, the same situation, but basal
angiohypertrophy is extreme, and the sprouting new vessel has crossed the
boundary into the Integra. The matrix
is populated by syncytial clusters, without any fibroplasia, density, or
consolidation. Left upper. A wider
view of a comparable specimen. angiohypertrophy is abundant across the base. Migratory histoprogenitor cells are
streaming throughout the tissue toward the Integra. Vascular ingrowth is occurring in several
areas, seen as the dark basophilic areas where angioblasts or angiocytes
create high cellular density. It is
around these new vessels that progressive proliferative histogenesis can
occur, and one can see in this image that around the basophilic zones, there is increasing density of the matrix due to
filling by cells and pink eosinophilic collagen. |
|
|
|
|
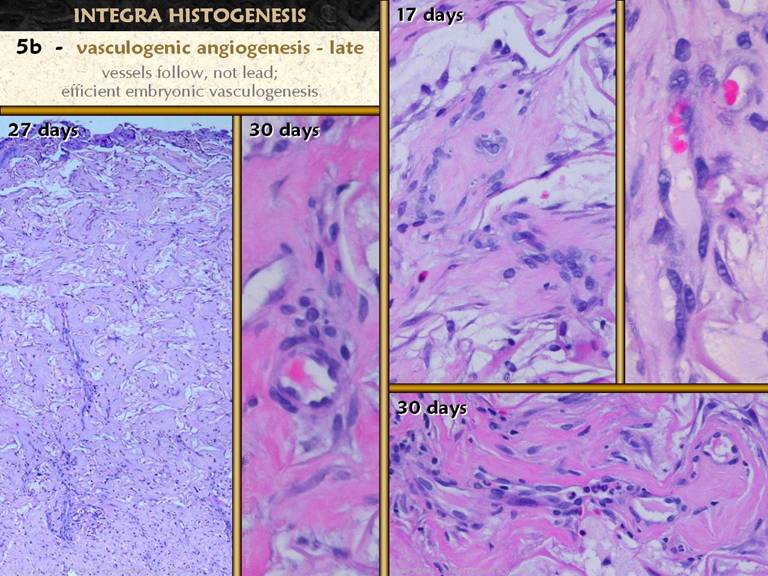
SLIDE 35The physics of vasculogenesis permit the early syncytial
clusters to develop throughout the matrix, up to a certain size. Once they reach the size where further
growth is limited by blood supply, then further histogenesis proceeds as a
wave or front, beginning at the base, adjacent to the wound, and moving
superficially toward the silicone surface.
This is because the source vessels are in the wound, and as they grow,
they first supply or relieve the lowest clusters, but they also establish new
vessels within the matrix which can then source angiocytes and vessels to the
upper strata. The physics of
vasculogenesis also dictate the caliber of vessels, with more central or
lower order vessels being bigger because they must admit more flow to a
larger domain of daughter vessels.
This means that as the wave of histogenesis advances through the
matrix, older earlier vessels near the base will get larger diameters (and
compensatory larger wall thickness, as cells in the vessel wall duplicate, to
normalize intercellular tension - recall LaPlace’s Law). Right center.
Fibroblasts and dense collagen are beginning to fill the matrix. Angiogenic cords are present, composed of
loosely organized angioblasts in a process of migration and
reorganization. Right upper, nearby
vessels are still somewhat disorganized compared to mature endothelial
capillaries, but they are sufficiently reorganized to have blood-conducting
lumens, evidenced by the erythrocytes. Center and right lower.
More advanced organization and maturation of recent vessels, showing
more mature coalescence of the cells into vessels, branching of smaller from
larger vessels, and the recruitment of new angiocytes to form onion-skin new
layers of cells to increase the wall thickness of larger diameter vessels. Left. A wide view of the full thickness of regenerating matrix. The entire matrix is now filled with tissue at one stage or another of maturity. Note the larger, longer, more obvious vessels deeper down, and the smaller thinner vessels higher up. Note too that although the deeper vessels are larger, that vascular density (vessels per voxel) does not vary much throughout the matrix.
|
|
|
|
|
SLIDE 36Pioneer cells get into the matrix,
settle onto the matrix as transitional cells, then transform into early
histogenetic syncytial cells. These
cells can form small clusters, until they get to a size that is limited by
vascular supply. They attract new
vessels from the underlying wound, which permits progressive cell function
and proliferation, leading to progressive histogenesis. This phase of progressive histogenesis is
characterized by the proliferation of more ordinary looking histioblasts and
fibroblasts and the deposition of fibrous collagen, both of which fill the
matrix with organized, continuous, consolidated material. Right upper.
Domains within the sponge, previously empty, are now filling to
capacity with cells and collagen. Note
that the process does not necessarily occur everywhere at once. Some domains are still empty or filled with
syncytial cells, while others are becoming collagenized. Angiogenic cords are present in the midst
of the organizing areas. Right lower. The
process is more advanced, with almost all spaces now filled with fibroblasts
and collagen. Middle. Note that
the process more or less advances as a front from deep to superficial, with
the deeper areas appearing comparable to the right-lower view, the mid zone
looking like the right-upper view, and the top of the matrix is still
relatively empty, having pioneer and syncytial cells. Left. A view which coincidentally or randomly has adjacent domains in various stages of this process. The top left domain has early non-vascularized cells at various stages. The top right domain is packed with cells, some syncytial and some more mature fibroblasts. The bottom domain is a little more advanced, with discrete areas of young, somewhat amorphous collagen appearing. The left middle domain is further along, with more diffuse collagen appearing throughout, displacing cells. The right lower domain is the most advanced, with collagen taking on a compressed, oriented, lamellar appearance, with entrapped cells likewise becoming long and flat, beginning to take on the morphology of mature final fibrocytes.
|
|
|
|
|
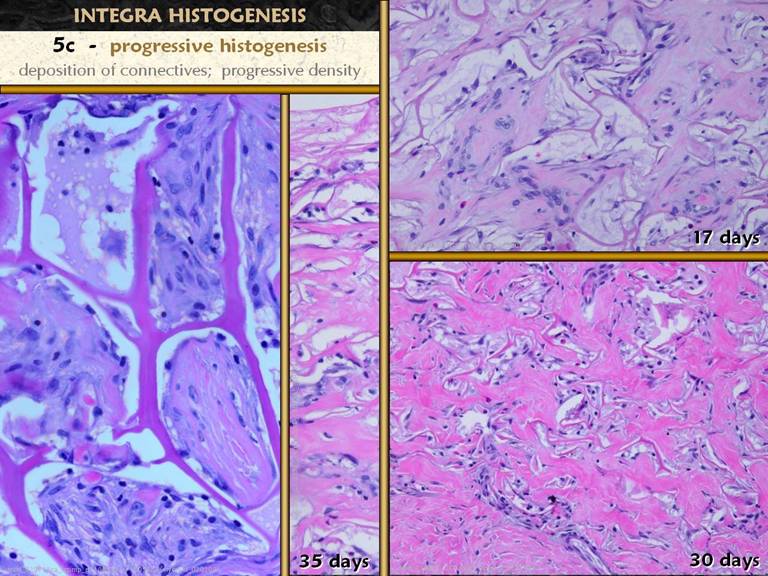
SLIDE 37Once blood vessels form in an area, progressive
histogenesis occurs, with each domain filling with fibroblasts and then
collagen. As this process becomes
thorough and complete, the collagen becomes denser, more fibrous and lamellar,
and fibroblasts become sparser and flatter.
Whether fibrocytes are diminished in number due to some type of
involution or disappearance versus all cells being present, but less
prominent due to flatter thinner cells within a bulkier matrix is an obvious
question. It cannot be answered with
certainty, but various features suggest the latter, that all young cells
survive as mature fibrocytes. Reasons
which suggest this include: the slow,
feedback controlled appearance of cells, a roughly similar cell count in any
domain regardless of early cellular versus late collagenized status, the
absence of histologic necrosis, cell ghosts, cell debris, inflammatory and
reactive cells, or any other sign of apoptosis or phagocytosis, and (as will
be seen in subsequent slides) the progressive thickening of the matrix, which
confirms that collagen quantity is increasing relative to original domain
size and cell packing. As the matrix
becomes more densely collagenized and cells flatten and mature, the matrix
grossly takes on its “regenerated” appearance. When fully regenerated, the matrix has a
distinctive appearance which is quite different than normal scar. Whether or not the collagen type profile is
different between Integra, scar, and normal dermis and fascias is an
intriguing question which would further define the differences or
similarities between Integra and these structures, but this question cannot
be answered by simple light microscopy. Left upper. Latter
stages of regeneration. Cells are less
dense as collagen fills domains. The
process is staggered in time from one domain to another, such that the domain
on the right seems very mature and striate, whereas the left upper corner is
still fairly cellular. Notice how the
domains are quite expanded vertically due to collagen accumulation. Left lower. Top of
the matrix. Some domains remain
slightly cellular, but mostly the cells are flattening and the collagen is
becoming more lamellar. Note that
these domains remain somewhat flattened, as not enough collagen has
accumulated yet to expand them vertically. Right upper. A
domain showing nicely the progressive accumulation and lamellation of the
collagen, with the fibroblasts flattening into their mature final form and
position. A regenerated vessel within
the center is the blood supply to this domain. Photo. Integra
fully regenerated and ready for skin grafts, corresponding to what is seen in
the histologic images on this slide. Right center and lower.
While some domains are still cellular and regenerating, the
accumulation of collagen in some areas is robust enough to make fibroblasts
seem very sparse. Note how collagen and cells respect the forms of the Integra sponge and its domains. Aside from some vertical filling or expansion, the morphology of the sponge remains undisturbed, without distortion, compression, crumpling or any other deformation that might be expected if the wound was behaving like a scar, with dense cellular collagenization and contraction. |
|
|
|
|
SLIDE 38Left. A back wound
that was closed with Integra. The
upper image is shortly after skin grafting.
Within a short interval after, the skin is soft and highly
compliant. It moves and wrinkles and
can be easily pinched and manipulated.
If this was normal scar (including conventional skin grafts), wound
contraction and stiff fibroplasia would render the skin non-compliant and
make this kind of motion impossible. Micro images. These
images are shown at various times after placement of Integra, beginning at
the end of the early histogenetic process when the matrix is filled, and
extending outward 4 years. As already noted, cells and collagen respect and conform to the
Integra, rather than subjugating the Integra matrix to fibrous replacement or
distortion due to contraction. While
these stains and images do not explicitly rule out the presence of
myofibroblasts, there is not the least telltale sign of any myofibroblast
activity. The absence of contraction and the high compliance and pliability of regenerated Integra seems to have two and maybe three origins. The first and passive reason is the lack of scar, i.e. that dense, contracting, tightly compressed, highly compacted and directional mass of collagen that forms during normal wound repair. A second possible reason, remaining to be investigated, is that the collagen types that appear in the Integra are different than what appears in scar. The third, the active reason, is that the architecture of the regenerated Integra may explicitly permit compliant motion. By having the matrix divided into domains by the Integra sponge, continuity of the fibrous tissue over long distances is prevented, and the conformity of the tissue to the spongy domains means that tensile vectors are distributed broadly in space, rather than along one direction, both factors favoring a more compliant material, even if the collagenous material itself is inherently stiff. |
|
|
|
|
SLIDE 39Normal open wounds can epithelialize to closure by
themselves, but skin grafts are a common therapeutic method to hasten this
endpoint. Integra is closed by skin
grafts. For all of these scenarios,
the basic biology of reepithelialization is the same. Newly arrived keratinocytes must reorganize
themselves into a laminated structure with a well formed basal layer (stratum
germinativum). A basement membrane
forms, created by the basal cells themselves.
A lamina propria, the papillary dermis, must form underneath as a
service layer to provide circulation and other support functions. Papillation occurs as the basal layer
expands to a size that can source the cells needed for a dynamic stratum
corneum, and to maintain the geometry of adequate blood supply. If the skin graft does not completely take,
then open areas can continue closing by natural epithelialization (proliferation
and migration of keratinocytes), after which the same epidermal maturation
events continue. These events are
governed by the epidermis itself, and they occur
independent of whether the grafts went on a normal wound module wound or
Integra or any other tissue. Left. Integra was
used to close a transtarsal amputation.
The Integra is healthy, but the skin graft did not completely
take. With basic hygienic wound care
to keep the wound safe, epithelialization to closure continues by itself. Center and right top.
Fully regenerated Integra ready for a skin graft, the close up showing
the surface upon which the skin graft is placed. Right center. Eight
weeks after skin grafting, the epidermis is adherent to the wound. The basal cells are disorganized and
dystrophic, and the stratum spinosum is also disordered, reflecting a graft
still trying to “get on its feet” after grafting. Right lower. Another view of the same patient at the same time, showing a slightly different more mature area. Basal cells in this view are still squamous rather than cuboidal or columnar, but otherwise healthy and more organized. A basement membrane is visible at higher resolutions (inset). A thin fibrous layer is present between the Integra and the epidermis, the beginnings of the papillary dermis. Only as this more completely forms will the epidermis get papillae and its own subepithelial circulation. |
|
|
|
|
SLIDE 40Integra undergoes a maturation process. The three phase of maturation are
conceptually similar to the maturation of a normal wound, but with some important
differences. In a regular wound, the
first phase of maturation is mesenchymal consolidation. Absent epithelial closure, the wound module
would continue proliferating, but when the wound is closed, the different
strata of the wound, reflecting different times of the wound, all come up to a
common level of early completion, forming the young scar. In Integra, the first phase of maturation
is likewise a consolidation to a common and nearly complete state of
histogenesis. Center. Regenerated
Integra at 4 weeks. The material seems
properly regenerated by gross observation (biopsy taken at the time of skin
grafting), but the microscope reveals that active post-syncytial histogenesis
is still taking place in the upper matrix, characterized by abundant cells
and early collagen (see the zoom in, left lower). The deeper layers of the matrix a re more
mature, with increased collagen, flattened cells, and reduced cell
density. Over the ensuing few weeks,
all levels of the matrix will grow to completion, until all layers look the
same (right two panels). In the middle
view, note that although collagen and cell density appear to be somewhat
uniform, that residual pale basophilia in the upper levels reflects residual
immature elements. Note that unlike a normal wound module, which will
continue proliferating until closed by epithelium, that
Integra regenerates only to the point shown here, and then no more. The risk of open Integra, without silicone
or epithelium, is that inflammatory wound healing and a wound module will
develop on the surface of the open Integra, but the Integra itself can only
regenerate until the matrix is filled and consolidated, and that’s that. These findings may explain a common undesirable property of Integra, the “disappearing skin graft” trick, in which a graft is placed on apparently healthy regenerated Integra, the graft looks good for a week or two, and then the graft starts to disappear. Second skin grafts usually take without problems. The photos illustrate this. A longstanding leg wound (A) is excised and closed with Integra (a 36 year old man with atherosclerosis, venous disease, diabetes, hyperlipidemias, and a hypercoagulable disorder). At 4 weeks, the Integra looks properly regenerated (B). The first set of skin grafts looks healthy at one week (C), but then it completely disappeared. A second set of grafts (D) is almost fully healed without loss. Why? At four weeks, an average or typical time to place the skin grafts, when the matrix is usually seemingly regenerated, there is still cellularity and incomplete histogenesis at the top level, where the skin grafts are to placed. Vascularization may not yet be sufficient, or it may be sufficient for the active fibroblasts, but not sufficient to support both populations of cells. Even if it is sufficient, the regenerating tissue may not be mature enough, inadequate collagen, fibronectins, or laminins for example, to permit stable adhesion of the graft. Waiting a few more weeks until the matrix is more thoroughly regenerated may be necessary to get the skin grafts to adhere. |
|
|
|
|
SLIDE 41The maturation of the epidermis during Integra reconstruction is identical to what occurs after ordinary skin grafts and wound reepithelialization. This includes stabilization of the epithelium, resumption of a columnar basal layer, normal development, proliferation, maturation, and turnover of the upper strata, formation of rete pegs, and formation of a papillary dermis with a subepithelial vascular plexus and papillary vascular tufts. The photos show the gross appearance of the foot skin graft as it fully matures. The various events are observed histologically. The center and lower right images show very nicely the new papillary dermis, created and regulated under control of the epithelium, with its subepidermal plexus arising from the reticular dermis of fully mature and persistent Integra. The upper right shows the blood supply to the epidermis, the papillary tufts. Right middle is a tangential section through the lower epidermis, showing mostly basal cells and acanthocytes, but also crosscutting the papillae, confirming that papillary distribution and density is normal. |
|
|
|
|
SLIDE 42The important differences between normal wound maturation
and Integra maturation concern the final stages. In a normal wound, excess cells and
proteins of the earlier proliferative process are dismantled and resorbed,
“overshoot then involute”. The early
scar, which was densely cellular, hypervascular, and excessively
collagenized, now starts to modify itself, thinning and remodeling itself
until eventually it achieves a structure somewhat comparable to normal dermis
or fibrous fascias. This process
occurs during a period of months to years. In distinction, no such process occurs in Integra. There is no resorption nor involution. The matrix builds to a model of mature tissue, then stays stable. In late mature Integra, the regenerated matrix is no different in appearance than it was at 2 to 4 months. From the point of matrix consolidation (left, right upper), the Integra simply remains as is long term (other images), with only very slow modification of the tissue. In the right middle and lower images, the matrix has lasted a very long time, but matrix resorption is occurring. In its place remains a loosely bundled wavy collagen which is not too different than its architecture with the matrix intact, and which is not too different than normal dermis. |
|
|
|
|
SLIDE 43Four views of the full thickness of the material at
various times after placement. These
are wide angle views, permitting a good view of the host-matrix interface and
the spatial distribution of local variances in the regeneration process. Left upper. At five
days, there is no physical connection between wound and Integra. angiohypertrophy
is evident, and progenitor cells can be seen streaming through tissue to
enter the matrix. the
matrix is relatively uniformly peppered with pioneer cells, and some early
syncytial clusters can be seen. Right upper. At 10
days, angiohypertrophy is substantial.
Streams of entrained cells are moving large numbers of cells into the
matrix. The upper half of the matrix
is no different than the 5 day view, with pioneer cells and a few syncytial
cells. Syncytial clusters can be seen
at mid matrix. In the lower half of
the matrix, early clusters have given way to domain-filling cellular
proliferation. Proliferation is most
dense closest to hypertrophied source vessels, where vasculogenesis into the
matrix first occurs. An actual
physical or anatomical connection of matrix to host is just beginning. Left lower. At 18
days, the process is advanced. There
is a firm fibrous connection of matrix to host. Well developed blood vessels have entered
the matrix, and matrix-filling histogenesis is now occurring in the mid and
upper layers of the matrix. The lower
layers have dense filling with cells and collagen. Empty matrix and syncytial clusters are
still present in the topmost stratum. angiohypertrophy is still present, but lessened, and new
vessels bridging between host and matrix are very well organized and clearly
delineated. Right lower. Advanced stages of regeneration. Although there is a lot of fixation artifact creating false empty spaces, the matrix is mostly filled with tissue (some domains may still be empty at the top). The regenerated tissue is largely eosinophilic due to collagen, without the intense basophilia due to dense cellularity. Perivascular angiohypertrophy is subsiding, and host or substrate anatomy is returning to normal. Collagen binding of substrate to new tissue in the matrix is advanced, and mature vessels bridging the interface are obvious. Note that the lower parts of the matrix are thicker, more expanded from more collagen, whereas the upper strata are flatter where there is less collagen, confirming that the matrix does expand vertically, getting thicker and more voluminous with progressive histogenesis. |
|
|
|
|
SLIDE 44Similar comparison views, vertical rather than horizontal,
giving a better sense of the timewise development of the new tissue. Note that the “##days” designation simple
documents when each specimen was taken, and is not to be interpreted as being
a strict timescale of histogenesis and regeneration. The times shown do accurately reflect the
general process and times that occur, but there are variances from patient to
patient, time to time, place to place, and even one millimeter to the next in
any specimen. The “17 days” specimens
from two different patients show two close but different phases of the
regeneration process. A. 5
days. The matrix is empty of proteins,
glycans, and any other formed substance.
Early pioneer cells sparsely populate the matrix, independent of
distance from the host. There is no
physical connection of matrix to host.
Some of the cells are adhering to the matrix, entering their
transitional phase before becoming actively proteogenic histogenetic
syncytial cells. B. 13
days. Cells have transformed into
syncytial histioblasts, and clusters of such cells are present. There is still no protein or glycan
matrix. The clusters sit in a
presumably serous medium, capable of functioning with oxygen and nutrients
that diffuse from vessels in the host, but reaching their limits of growth and
activity until direct vascularization of the matrix occurs. C. 17 days,
left. The matrix is populated by large
branched (pseudopods) syncytial histioblasts.
At the interface with the host wound, angiohypertrophy is evident, and
migration and ingrowth of cells from host vessel into matrix can be
seen. Surrounding this zone of
vascular infiltration, cell density is increasing in the sponge, and early
organized collagen is appearing. D. 17 days,
right. The process of vascular
infiltration and progressive histogenesis is now in full swing. There is a firm physical connection between
host and Integra. angiohypertrophy
is still evident at the base, and long new vessels are snaking up into the
matrix. At the lowest levels,
generally eosinophilic pink, collagen deposition is dense. In the upper half, just above the large
vessels, is a basophilic zone of small capillaries supporting dense
proliferating histioblasts which are just starting to make collagen. The upper layer or non-staining stratum has
syncytial cells, pioneer cells, and empty domains of the sponge. E. 30
days. The same process continues,
progressing up to the top of the matrix.
Vascular ingrowth is now evident throughout the matrix, with larger
conducting vessels rising high enough to permit substrate supply to the top
strata. Basal angiohypertrophy has
subsided, because histoprogenitor cells at this level no longer feel the
effects of proliferative cytokines coming form the zone of active
histogenesis which by now is quite far away.
The lower areas of the regenerated matrix are increasingly
eosinophilic, as collagen accumulates and matures, and fibrocytes become
thinner and less active. In the upper
half, there is still a purple basophilic balance to the color, due to a
higher density of cellular cytoplasm and nucleoplasm, and a relative lack of
collagen. This zone corresponds to
what was starting in the basal area in panel C, and what was occurring in
panel D above the middle. F. 42
days. The entire matrix is now filled
with collagen. Cell proliferation in
the host is subsided, and cell density throughout the matrix is
diminished. Vascular density is
uniform throughout the regenerated matrix.
However, note that there are still differences between the upper and
lower strata. Below, collagen is
pinker, denser, more organized, whereas above, there is still a relative
basophilia, , and collagen is less dense or
organized. G. 70
days. The process is now almost
uniformly complete throughout the matrix, with only a slight residual
basophilic tint in the topmost zone.
New vessels crossing the interface, and the
tissues of the host have returned to normal appearance and cell density. Note how the matrix gets progressively thicker as collagen fills up the sponge domains. Papillomatosis of the Integra never occurs, meaning that the material is not expanding tangentially, only vertically. |
|
|
|
|
SLIDE 45Another panel of progressive images, showing what happens
after skin grafts are applied. A. Early. The skin graft is firmly adherent
to the matrix. Acanthocytes
predominate, and the basal layer is still reorganizing into a correct stratum
germinativum. The graft sits directly
on regenerated Integra matrix. There
are no new mesenchymal elements that were not there when the graft was
placed. B. Later. A normal layer of basal cells has reformed,
functioning correctly as evidenced by acanthosis and early papillation. A papillary dermis, which is triggered and
governed by the epidermis, is beginning to form, a cellular and collagenous zone
of new tissue between matrix and epithelium.
angiohypertrophy is seen at the top of the
Integra, because these vessels now become the source of the the subepidermal
plexus and papillary tufts that nourish the epidermis. C, D, E. Mature, 1
- 4 years. The epidermis is mature in
all respects, including mature papillation with rete pegs. A normal papillary or subepidermal dermis
has fully formed, containing mature subepidermal and papillary blood
vessels. The original Integra sponge
is still present below, perhaps thinning out here and there, but still having
an overall normal appearance, without evidence of contraction. Gross architecture of the regenerated
Integra, which is the new reticular dermis, is quite similar to normal dermis
in terms of the organization and density of collagen fibers. Even after the matrix starts to disappear
(E, right), the collagen retains its irregular organization, continuing to
look more like the mechanically advantageous collagen architecture of normal
dermis rather than the tight, dense, non-compliant architecture of normal
post-inflammatory scar. Slow disappearance of the original collagen-gag matrix is presumably by hydrolysis alone, or hydrolysis plus low grade non-specific (perhaps even non-enzymatic) proteolysis or glycolysis. There is never any evidence of a reactive, inflammatory, defensive, or destructive cellular response to the matrix, and the very slow disappearance of the matrix argues against any type of specific collagenase or other protease. |
|
|
|
|
SLIDE 46The process of Integra histogenesis has now been
examined. There are distinctive events
which are in some ways analogous to those of normal inflammatory wound
repair, but with important differences in detail and sequence. Some of the events of these two processes
are distinctly different. Zones and
phases of regeneration can also be delineated, just as for inflammatory
repair, but there is very little if any similarity between the two systems
regarding their stratigraphic anatomy. The biggest distinction between these two systems, from a
physics or systems point of view, is that Integra histogenesis is a process
of steadily building up a mature tissue, beginning with nothing, and
asymptotically approaching the final model.
Recall that with inflammatory wound repair, the response is a rapid
over-attraction, over-production, over-accumulation of cells, gag’s, and proteins.
Once the wound is epithelialized and closed, the process subsides, and
then the scar matures, gradually and asymptotically modifying itself back to
a structure that is similar (but rarely identical) to normal dermis or
fascia. This process of “overshoot
then involute” is the basis for scar’s many undesirable properties. The acute open loop auto-amplifying
“overshoot” phase is a process which evolves in days to weeks. Subsequent maturation and modification back
toward reference anatomy is a process which takes months to years. In comparison, Integra histogenesis, with
closed loop controls, steadily builds toward the reference anatomy, evolving
more gradually, but completing more quickly, in a period of weeks to
months. (Open and closed loop controls
and other dynamical differences between wound healing and Integra
histogenesis will be considered in the next section). |
PART 3: Inflammatory Repair and Integra Histogenesis
SIDE-BY-SIDE
|
|
|
|
|
SLIDE 47This presentation began with the statement that Integra
has superior properties compared to normal wounds and scars, and the reason
for this is that Integra heals by a method comparable to embryonic
histogenesis rather than by the normal inflammatory wound module. Normal wound healing and Integra
regeneration have been presented in detail.
They next few slides will be a side-by-side review of these two
processes, and then the nature of embryonic histogenesis and its similarities
to Integra will be explained. |
|
|
|
|
SLIDE 48Review and side-by-side comparison. 0. Injury is
recognized, triggering the process of repair.
Inflammation induces the process of repair, but inflammation must also
subside or be sequestered before repair gets fully underway. With Integra, injury induced inflammation
and its resolution are irrelevant, because the wound must be managed to
control all pathology and inflammation prior to closure, and the wound is acutely
excised when Integra is placed. An
excised wound means that, while the Integra is being placed on injured
tissue, it is nevertheless normal tissue, without inflammation nor
inflammatory wound healing nor any of the cells and chemical mediators that
govern that process. 1. Inflammation is
a crucial host defense process. The
outer inflammatory layer of the wound forms a barrier below which later
phases of repair can occur.
Inflammation induces monocyte-macrophage transformation which
regulates the subsequent repair. With
Integra, the entire material is the sequestered space in which histogenesis
can occur. Acute inflammation
initiated by the surgery is completely arrested by the Integra. Blood borne cells do not accumulate, and
there are no monocytes nor monocyte transformations. |
|
|
|
|
SLIDE 49Review and side-by-side comparison. 2a.
In a normal wound, dense accumulations of inflammatory cells
appear at the boundary between viable and non-viable tissues, where the
macrophages have an afferent function to remove debris and necrosis, recognized
clinically by eschar separation. In
Integra, there is never inflammation, and there is never an accumulation of
reactive or defensive cells, nothing in any way analogous to eschar
separation. The only cells which do
proliferate are the cells of histogenetic regeneration, and they appear in a
sparser, more regulated or metered way. 2b.
In a normal wound, the efferent macrophage function is to
stimulate local responder cells to begin making new vessels and fibrous
tissue. In Integra, there clearly has
to be some type of recognition, but it is not by
macrophages nor other obviously blood borne cells. It appears that cells in the substrate
tissue, some type “patrol” cells, derived from angiocytes or tissue
histiocytes, find the matrix, and it is the recognition of the matrix alone
which triggers subsequent events. In
the absence of specific identification of the origin of the pioneer cells,
one could even postulate that they are blood borne lymphocytes, true or
not. If hypothetically true though, they
are leukocytes with a crucial difference:
they arrive randomly and in low numbers, in the normal course of
exiting the blood and patrolling the tissues, coincidentally finding the
matrix, maintaining primitive histogenetic pluripotentiality, rather than
being cytokine-triggered responder cells, arriving in large numbers,
transformed and ready for more targeted jobs, destined only to do their job
and leave before histogenesis starts. |
|
|
|
|
SLIDE 50Review and side-by-side comparison. 3 / 3a. With both
inflammatory repair and Integra, something recognizes the injury, and some
type of early responder cell appears which then regulates the subsequent
histogenetic process. The general
overall phases of this histogenesis are comparable between normal wounds and
Integra, but in each subphase, there are important sequence inversions
between the two systems. In
inflammatory wounds, the first histogenetic event is the appearance of ground
substance, created by macrophages or other acute cells, creating the “ether”
in which histogenetic cells can survive, migrate, organize, and
function. The appearance of a ground
substance rich in carboxylated and sulfurated glycosaminoglycans must precede
the appearance of locally derived histogenetic cells. In Integra, the Integra material and sponge
itself seems to serve as a suitable matrix that cells recognize as
“home”. Thus, the early cells can
initiate their own histogenetic processes without an additional syrupy space
filling aminoglycan soup. As they
start to proliferate, they do begin to make aminoglycans, but only in the
small regulated amounts needed to allow new cells to occupy the interstitial
spaces. Early histogenetic cells
precede GAGs (sequence inversion), and GAG production is more regulated. 4a / 3b. In both
normal wounds and Integra, the process of histogenesis depends on the
proliferation and organization of vascular cells and fibrous cells. The order of appearance of these cell lines
is another sequence inversion, but in both cases, these two cell lines are
being supplied by local mesenchymal progenitor cells responding from layers
subjacent to the wound or Integra surface.
What are the origins of these cells?
In both cases, the cells are coming from underlying blood
vessels. The vessels that respond and
supply can be large muscular arteries, large adventitial veins, or the
tiniest capillaries. The angiocytes do
not seem to care where they are. They
can be endothelial or medial or perivascular.
In observing the proliferation of cells from old vessels and their
reorganization into new vessels, one gets the sense that an angiocyte is an
angiocyte. Cells may appear different
due to spatial and mechanical and geometric circumstances, but they all seem
to maintain a primitive pluripotentiality that permits seemingly immortal
replication of the cell line to make new vessels and fibrous tissue. The two bottom pictures show an identical response to
histo-proliferative stimulation. The
only thing that gives away the identity of the two situations is that one has
neutrophils streaming from the vessel in response to continued inflammatory
stimulation, and one has none. Look
past the leukocytes, and what do you see?
Angiocytes have become larger, with more cytoplasm and bigger
nuclei. The cells are transforming
from flat and lamellar to round and more individualized. Medial and adventitial layers of cells are
all responding, and equally so are the intimal (endothelial) layers. Some cells are becoming spindled and
elongated, preparing to migrate, and some are already migrating. Some remain in place but are undergoing
mitosis to source new cells. As the
process reaches its peak, the hypertrophy and thickening of the source
vessels and the shedding and streaming of cells into the wound or Integra
becomes quite dense. Throughout this presentation, these cells have been
referred to as the following: 1 -
angiocytes, meaning either any general vascular cell, or specifically a
mature functionally stable non-proliferative vascular cell; 2 - angioblasts, meaning either those cells
which have become stimulated proliferative source cells, or those cells which
have stem-like latent responder potential; 3 - angiopericytes, to designate
non-endothelial non-specific adventitial or medial cells (as opposed to
mature smooth muscle cells as might appear in larger arteries). This choice of words is based on the basic
etymological rules of medical terminology, but keep in mind that the meanings
of these words as used throughout this presentation are partly contextual,
and this nomenclature may cross paths with terminology used by other authors
studying various other biological models. Key to understanding both normal wounds and Integra is to
recognize that there is only one source of histogenetic cells. How and when these cells respond, how they
themselves organize and interact, and what type of biological output they
create are all a consequence of the inflammatory-macrophage-ground substance
milieu versus the non-inflammatory-Integra-pioneer cell milieu. This is another example of the biological,
physical, systems principle of parsimonious self-organization. |
|
|
|
|
SLIDE 51Review and side-by-side comparison. 4b / 3c,4a. In a normal wound, the first histogenetic
cells are angioblasts. They are seen
in the image above as individual migratory spindle cells streaming from
vessels underneath where they originate toward the macrophages which are
issuing chemotactic cytokines (and interspersed with acute inflammatory
cells). They are seen in the image
below in a more organized state where the cells have either remained coherent
or have reassembled into new lumenized blood bearing vessels. In Integra the first histogenetic cells are the pioneer
cells cum syncytial histioblasts.
These cells do not profusely and indiscriminately overrun the matrix
the way early angiocytes stream into the inflammatory wound. Instead, only relatively limited numbers of
pioneer cells appear initially, and subsequent
syncytial cells form localized clusters as they arise from the progenitor
cells. 4b / 3d,4b. No cells or tissues survive or function
without adequate blood circulation, and this is especially true for the
highly metabolic proliferative healing wound, for growing embryonic tissues,
and for regenerating Integra. In a
normal wound, excessively dense angiogenesis leads, preparing an environment
for subsequent fibrous cell proliferation.
In Integra, primitive fibrous cells appear first, but absent a
concomitant circulation, there are limits to how much these early syncytial
clusters can grow. When the size and
metabolic load in these clusters exceeds supply, these cells then respond to
make angio-stimulatory cytokines. This
regulated process is identical to that of normal embryonic
vasculogenesis. It is a systems level
process governed by basic mathematical and physical principles, and mediated
largely by oxygen and angiogenic cytokines, in which existing blood supply in
any locale can support only a certain number of cells. As each syncytial cluster starts to attract
new blood vessels, conditions are established that permit further mitosis,
cell aggregation, and export proteogenesis, and the matrix space now starts
to fill completely with cells and collagen.
The upper image shows a syncytial cluster at the limits of
proliferation absent new circulation.
The lower image shows a wider view with many such independent
clusters. The one in the lower left
area, adjacent to the host, is already establishing new local circulation, as
evidenced by the deposition of collagen and a wider zone of histogenesis. (The numerous red blood cells are a
bleeding artifact from taking the biopsy.) |
|
|
|
|
SLIDE 52Review and side-by-side comparison. 5a / 5a,5b. In a normal wound, fibrogenic cells appear
only after angiogenesis has created a dense hypervascular network. Histoprogenitor cells that are to be
committed along fibroblasts lines now start to proliferate and function
between the new well-established vessels.
In the image above, vessels are visible at upper right and lower left
corners. In between, what was
previously GAG filled “empty space” is now filled densely with young
histoprogenitor cells. They are round
or elongate, with large nuclei, with pale pink color attesting to the
appearance of collagen. In the image
below, these cells are in a slightly more advanced state. The cells are becoming flatter and more
elongate, starting to develop a sense of laminar clustering and directional
orientation. Cells are becoming more
widely spaced as collagen accumulates. In Integra, vessels follow histioblasts. Revascularization is actually the keystone
between early histogenesis with syncytial histioblasts and later mature
fibroplasia. In the image above,
vascular and perivascular hypertrophy is obvious in vessels underlying the
Integra, and growth of a new vessel into the Integra is clearly seen. Trains of darkly basophilic cells attest to
other angiogenic cords progressing through the matrix, not in the diffuse
dense front typical of inflammatory angiogenesis, but in the controlled or
regulated isodensity way characteristic of aiming at discrete stimulatory
loci. Note also that around the vessel
entering the matrix, that the matrix itself is starting to consolidate,
filling with collagen as fibrous cells start to function fully in the
presence of blood supply. while collagen deposition and consolidation is starting,
cell density remains low, never appearing like the dense cellularity
characteristic of the comparable phase of inflammatory repair. In the image below, the same events are a
little more advanced. Diffuse
basophilia across the surface of the original wound is due to intense
angiohypertrophy and proliferation of histoprogenitor cells. The upper layers of the matrix are still
sparsely cellularized, with pioneer cells and syncytial clusters, but
angiogenic cords can be seen working their way throughout the matrix. In the lower layers of the matrix,
angiogenesis has established mature vessels, and around them, collagen is
building up. Note that unlike an
inflammatory wound where cells and collagen completely fill the space, that
Integra histogenesis continues to occur in discrete zones, filling some
matrix spaces but leaving adjacent ones open, and never with the same high
concentration of fibrous cells. |
|
|
|
|
SLIDE 53Review and side-by-side comparison. 5b / 5c,5d. In a normal wound, once a population of
fibroblasts is established, the wound becomes progressively filled with
collagen. The flattening and lamellar
orientation of the fibroblasts continues, leading to thick fibers
characteristic of a normal scar. In
the image above, active fibroplasia is at its peak. Cells are dense, and the cord like
architecture of the fibrous bundles is well organized. There is still a lot of background
basophilia, not the cell nuclei which are obvious, but the paler purple haze
between collagen bundles representing cytoplasm which is still enlarged and
actively proteogenic. As the process
moves to more complete phases, image below, mature fibrous collagen entraps
the numerous fibrocytes which are now getting smaller and gearing down from
their peak proteogenic activity. In Integra, the appearance of blood vessels allows the
early histogenetic cells to further function, becoming more normal appearing
fibroblasts. The subsequent process of
fibrogenesis is comparable in some ways to inflammatory healing, but
different in many ways. In the image
above, angiocytes of a new vessel remain large and loosely organized, either
still condensing into a cylinder, enlarging the cylinder for greater
capacity, or sourcing cells for new vessels, but it is functional and
carrying blood. Around it, histogenic
cells are looking less syncytial and more like fibroblasts, thinning,
flattening, developing some lamellar or directional orientation, and
producing more collagen. Some cells
remain enlarged and syncytial and some matrix spaces remain unfilled. In the image below, the same thing is seen
at a wider view. On the right and at
right center are two organized blood vessels.
Take them out of the picture, and cell density is somewhat sparse, not
at all like the cell density during active fibroplasia in a normal scar. Within different locales, the process is in
various stages of evolution, everything from pioneer cells to syncytial clusters
to mature fibroplasia. Collagen is obviously accumulating and filling space,
but compared to what is seen in a normal scar, the collagen appears
different. In Integra, it appears finer, more fibrillar, almost amorphous in some places,
and while it can be lamellar and oriented over short distances within an
individual matrix space, there is no general orientation or directionality
throughout the space. |
|
|
|
|
SLIDE 54Review and side-by-side comparison. In Integra, wound contraction does not occur. One can infer that myofibroblasts therefore
do not appear, but even if more refined studies were to show that some
Integra fibroblasts do indeed express muscle proteins, there is no
demonstrable effect of their presence.
In the image above, one year after Integra placement, the matrix is
undistorted. If scar contracture were
to have occurred, there would be some type of deformation of the matrix,
compression, crumpling, stretching, flattening, lengthening, etc. Instead, there simply is no deformation,
period, neither acutely nor long term.
This property of Integra is also used therapeutically, to avoid or
correct scar contraction where normal wound healing would otherwise cause
disfiguring or disabling contractures. In Integra, the same is generally true. Integra reconstructs and regenerates a
lamina of mesenchymal tissue. This
lamina or “neodermis” sequesters the underling original wound and anatomical
structures, but as a mesenchymal structure, it is not fully healed until it
has itself been topped off by epithelium.
In principle epithelialization could be allowed to occur the natural
way, by migration from the perimeter, but in practice, it is restored by
surgical grafting. Although natural
epithelialization could be done in principle, various factors make this
undesirable. The first is that, in
normal wounds, contraction is usually responsible for the greatest
percent-of-area contribution to closure, minimizing the load on the
epithelial cells. Because Integra does
not contract, and because most Integra wounds are fairly sizeable, epithelial
ingrowth to closure would be prohibitively long. Also, unepithelialized Integra sometimes
seems to want to stay that way, but generally, small areas where surgical
grafts did not take at first go on to re-epithelialize like any other open
wound. The image shows a recently
placed skin graft over fully regenerated Integra. The basal layer of cells is not yet normal,
but is getting reorganized into a functional stratum germinativum. An epidermis-induced lamina propria, the
papillary dermis, has just barely started to appear, as a thin layer of cells
and collagen between the epidermis and the Integra. |
|
|
|
|
SLIDE 55Review and side-by-side comparison. 8a.
Once a wound is fully epithelialized, there are no longer any
foci of acute inflammation, all triggers to inflammatory repair are
eliminated, and all zones of the wound can catch up to each other. Once the triggers to angiogenesis and
fibroplasia are gone, the wound becomes relatively stable, and subsequent
changes occur slowly. The scar is like
any other connective tissue in that there is a slow and gradual turnover of
connective proteins and cells, the tissue gradually remodeling itself in a
process that is observable only at intervals of months or years. Because the normal scar is excessively
vascularized and collagenized, the gradual remodeling of the wound eventually
eliminates the excess elements until the ultimate mature tissue, months or
years later, looks much like normal dermis or fascia. In the first phase of this maturation,
shown in the images above, the various zones of the wound catch up to each
other, resulting in a final consolidated fibrous model having thick collagen
bundles and numerous but mature fibrocytes entrapped in the scar. In Integra, There is likewise a process of consolidation
in which the upper layers complete the regeneration process that first
started and completed in the lower layers, until the entire matrix, top to
bottom, looks completely uniform. The
image above shows healthy regenerated Integra at this point. 8b.
The next aspect of this late phase is the maturation of the
epidermis and the appearance of the papillary dermis. The events of this process are intrinsic to
the epithelium and epithelial-mesenchymal interactions, and as such, the
process is identical between normal wounds and Integra. As the epithelium matures, it gets thicker,
more stratified between basal and keratin-producing layers, papillated to
acquire necessary basal cell mass and adequate blood supply geometry, and
more mature in its functional output of a stratum corneum. The lamina propria that services the
epidermis, the papillary dermis, forms as a collagenized and vascularized
layer on top of the original wound or Integra, beginning with the appearance
of an interlaminar (“subepidermal”) vascular plexus which sources the
papillary vascular tufts. |
|
|
|
|
SLIDE 56Review and side-by-side comparison. 8c.
In a normal wound, the last and longest phase of maturation is
the gradual remodeling of the wound which eliminates excess vessels and
collagen until the scar eventually looks nearly like normal dermis or
fascia. The kinetics of soft tissue
remodeling, chemical and cellular turnover, are normally slow, so the
maturation of the scar, from thick, stiff, non-compliant, congested, and
hypertrophic, to nearly normal occurs slowly, measured in months to
years. The images show young scar
(left) which has dense cellular collagen and numerous enlarged vessels. In a fully mature scar (right), the vessels
have all returned to normal size and density, and collagen bundles appear
wavy and more compliant. In Integra, slow maturation is also observed, likewise
over a period of months to years, the Integra slowly looking more like normal
dermis and fascia. The difference
between scar and Integra is that maturation and the approach to a normal
tissue occur from opposite sides of the fence. Early wound healing is amplified and open
loop, depositing an excess of cells and proteins, which once in place,
remodel slowly back toward normality, a process of rapid overshoot (days to
weeks), then gradual involution (months to years). In Integra, there is no overshoot, just a
gradual but persistent regulated build up to a reasonably normal tissue. This process occurs over weeks to months,
and while further maturation to an even more normal appearance continues for
years, the gross clinical features of healed Integra (appearance and
compliance) are present within the first few months, and they change little
after that. |
|
|
|
|
SLIDE 57Review and side-by-side comparison. These two views show a normal wound and regenerated Integra
side by side. The normal wound will
forever have a stratified architecture, with inflammation at the top and
fibroplasia at the bottom, until epithelium closes the wound. Integra, not having inflammation, and
healing by a fundamentally different mechanism, builds to a state of complete
regeneration, uniform throughout the matrix, and then that’s the way it
stays. |
|
|
|
|
SLIDE 58Review and side-by-side comparison. In this panel, Integra, wounds, and skin are compared side
by side. Normal skin is on either
side. Note that the reticular dermis
has collagen bundles which are thick and well organized, but also wavy and
compliant, with numerous clefts between bundles. Normal wound module and normal Integra,
when healed, are equivalent to the reticular dermis in terms of their
relationship to underlying original tissues and overlying epidermis and
papillary dermis. Note that early
Integra, even though skin grafted, has not yet developed a papillary dermis. The late Integra, with its mature epidermis
and papillary dermis, looks much like normal skin, with large but loose
collagen bundles comparable to normal dermis.
In contrast, a mature scar, healed by natural epithelialization over
wound module, is distinctly different, having thick, cellular, non-compliant,
dense collagen. The mature skin graft
is comparable to a regular wound, the epithelium having been supplied by the
surgeon rather the wound margins. In
this example, the scar under the old skin graft is looking a little more like
dermis, due to the longevity and maturation in the wound. |
|
|
|
|
SLIDE 59In this and the next slide, the wound module, the normal
process of inflammatory wound repair, can be seen side-by-side against
Integra. Left upper. Normal
inflammatory “granulation tissue” is growing out of the seam between two
pieces of Integra. In this gap, small
to the naked eye, but big to individual cells, the absence of matrix and
silicone pseudo-epithelial coverage is simply a normal open wound, and it
tries to heal the normal way - the wound module of inflammatory repair. Left lower. A
different patient, but a comparable situation, now grafted and over one year
old. The Integra reconstructed zone
looks somewhat different than normal skin, due to minor surface texture
irregularities, some minor pigment variegation, and absence of subcutaneous
adipose fascias, but otherwise the Integra is fully mature, soft, and very
similar to normal skin. This is
especially so when it is seen juxtaposed against the now epithelialized
granulation tissue scar in the middle.
This scar remains hypertrophic, stiff, and excessively vascular, quite
a distinction from the Integra, especially given that both areas started at
the same time. Right. A biopsy was
taken from normal healthy regenerating Integra at 6 weeks. The biopsy site was left open. Two and a half weeks later, a new biopsy
was taken, centered on and at right angles to the first biopsy. This second biopsy, shown here, captures
the interface between healthy Integra and a healthy normal wound (the first
biopsy site). Once healthy Integra is
injured, the open wound is an open wound, and it responds with inflammatory
wound healing (unless of course a new piece of Integra was used to close the
wound). The wound module of
inflammatory repair, with granulation tissue and scar, appears completely
healthy and normal in this image, just as it should. The Integra likewise looks exactly
normal. The distinctions between the
two tissues are obvious when seen side by side. |
|
|
|
|
SLIDE 60This is a side by side comparison of late Integra and
normal scar. The Integra was used to
cover a complex flank wound. This
specimen was obtained one year later during further tumor surgery. Kidney is in the left lower corner. Overlying the kidney is an area of normal
scar, and adjacent to that is one year old Integra. The scar is dense, flattened and
contracted, with thick unidirectional collagen bundles. Compare this to the more open, less dense,
more isotropic (non-directional) orientation of finer collagen in the Integra. |
PART 4: COMPARISON TO EMBRYONIC HISTOGENESIS
|
|
|
|
|
SLIDE 61The remaining slides will develop the theory that Integra
histogenesis is comparable to normal developmental embryonic
histogenesis. This will be done first
by focusing on the formation of the blood vessels (angiogenesis) and the
vascular networks (vasculogenesis) in these systems. The first distinction is in noting the different dynamic
interactions between angioblasts-vessels and histioblasts-fibroblasts. In normal inflammatory repair, vessels are
triggered by stimuli (blood borne macrophages) that are not
part of nor destined to be part of the ultimate new tissue. New vessels develop along a unidirectional
front, and they lead the formation of fibrous cells and proteins which depend
on the logistical supply provided by the new vessels. In Integra histogenesis, histogenetic new
cells, locally derived, proliferate first (syncytial histioblasts). These cells and clusters, which are the
source of angiogenic stimulation, will be part of the new tissue, and they
lead the vessels. They also form a
distributed or dispersed field of stimulation which means that new vessels do
not grow along a unidirectional front but rather distributed throughout the
field. These latter statements concerning
Integra histogenesis also exactly describe normal vasculogenesis during
embryogenesis. Left, top to bottom.
Vasculogenic dynamics during inflammatory wound repair. A.
Streaming angioblasts are visible in the subsurface GAG layer, moving
toward transformed macrophages. This
stimulus and migration occurs everywhere across the surface of the wound,
without variations in density nor spatial gaps in
activity, because the triggers to this activity, inflammation and monocyte
transformation, are homogeneously distributed. B.
At a lower level, organized vessels suspended in GAGs created a rich
overly dense vascular network which can now supply the needs of a soon-to-be
rapidly proliferating and highly proteogenic crop of fibro-histioblasts. C.
Histogenetic cells are now appearing between the organized vessels,
and young collagen is starting to appear.
D. These histioblasts assume a
mature functional status in the form of typical fibroblasts, numerous, dense,
spindle shaped, surrounded by increasingly condensed collagen. Remember, vessels lead fibrous cells,
growing toward a front of attraction of cells which are not local and will
not be part of the ultimate tissue. The process of inflammatory wound repair is open loop or
auto-amplifying in many ways, which accounts for the robust cellular
recruitment and highly proliferative nature of the process. With regards to the activation of
angiogenesis, the process is open loop.
This is because the controller which drives the load, the macrophages
which stimulate the angioblasts, is not inhibited by the load or its
output. Just because the new vessels
arrive at the stimulating macrophages does not mean that the macrophages turn
off. While the macrophages are
themselves controlled by something (the acute inflammatory mediators), with
regard to the blood vessels that they are attracting, the macrophages are
autonomous and independent. Because
the macrophage-angioblast interaction is open loop, the inflammatory process
does not care how many new blood vessels form, and the number is substantial,
far in excess of what is needed for normal development and tissue function. Right, left and top to bottom. Vasculogenic dynamics during Integra
histogenesis. A. Histogenesis starts with syncytial
histioblasts. Like monocyte-macrophages,
they stimulate and attract angioblasts, but the similarity ends there. These cells are locally derived, they are
the source of further “productive” cells (fibroblasts), and they will
contribute to and become part of the mature tissue. B.
Unlike macrophages which exist uniformly dispersed throughout the
upper inflammatory layer of a normal wound, the syncytial cells are dispersed
throughout the Integra matrix. They
form syncytial clusters which function autonomously, beginning to create
connective proteins, supplied by normal vessels within the underlying
substrate tissue, not needing new vessels until their metabolic activity
outpaces available supply. C. Only as the clusters exceed certain sizes
and metabolic requirements and start to compete with each other do they issue
angiogenic cytokines to marshal new vessels.
In this image, angioblasts and young new vessels emanate from
substrate vessels, reaching tropically toward the clusters that are
stimulating them. Because the points of
stimulation and attraction are discrete and dispersed, vessels do not grow
indiscriminately toward the top, but rather in an orderly way toward the
attracting locales. D. Only after new vessels reach the clusters
can these clusters resume their metabolic functions, giving rise to more
fibroblasts, and creating mature space-filling connective tissue which moves
outward around the original clusters, the various original independent
locales eventually becoming confluent. Unlike inflammatory angiogenesis, blood vessel formation
in Integra is a closed loop process with feedback and a system
reference. Angiogenesis is not
triggered until cell mass and metabolic load in independent locales exceeds
existing supply, and then angiogenesis is triggered, sending new vessels only
to locales where beckoned. Arriving
blood supply suppresses the further production of angiogenic cytokines by the
tissue cells, and only just the necessary number of new vessels appears. Attraction of new vessels by stimulating
histioblasts, and the suppression of that attraction by the arrival of blood
vessels constitutes closed loop control.
While this presentation has not looked at cytokines and individual
cell kinetics, nor have vessels or vessel densities been counted, gross and
histologic pictures of regenerated Integra compared to normal wounds show
these concepts, as will be seen in the subsequent two slides. More important, these observations are
completely consistent with the known systems-level physics and physiology of
vasculogenesis. The goal here is to show that Integra histogenesis is the
same as embryonic histogenesis. What
is known about the systems-level physics and physiology of angiogenesis is
based on normal embryonic growth and development. In that situation, what happens is that
individual locales of proliferating tissue trigger tropic angiogenesis only
as metabolic loads outstrip available supply.
Arrival of blood vessels suppresses further angiogenic stimulation,
and vascular density is only exactly what it should be to meet metabolic
load. Much of this system is
understood, for instance that various angiogenic cytokines can be involved,
but that VEGF is the predominant one; that all proliferating or growing
embryonic tissues make VEGF during active tissue growth; that the VEGF gene
is directly turned on and off by oxygen or its absence; that vascular morphology and density are
highly organized and efficient, exhibiting the property of “locality” where
vessel growth is governed independently by distributed locales of active
growth. This description sounds
identical to what happens during Integra histogenesis. |
|
|
|
|
SLIDE 62This slide serves as support to the argument that Integra
vasculogenesis is conceptually and dynamically similar to embryonic
vasculogenesis, by demonstrating what is known about normal vascular
development. The VT (Vascular neT)
model of angiogenesis is a numerical model of the non-linear physiology of
embryonic vasculogenesis. The model
has just four inputs, a “growth model” that governs how the host tissue grow,
and three intrinsic angiogenic parameters:
the “ischemia threshold” which governs how far away from the vascular
network a cell in the tissue must be to “feel” the ischemia and trigger angiogenesis
by cytokine stimulation; the “reach”
which governs how far from the network to the stimulating cell a new vessel
grows; the multiple sprout multiplier
or “anastomosis” factor which governs the number of new vessel sprouts which
respond to angiogenic stimulation. The
model is based on the physics of diffusion and the known physiology of
angiogenesis and angiogenic factors.
The natural process is closed loop and tightly controlled, and the
model, which mimics the natural process, is iterative and non-linear. While beyond the scope of discussion of
this presentation, this model can accurately recreate the morphology and
engineering parameters of any vascular and other networks in a diverse array
of biological systems. The images
shown here illustrate the model. Left upper. A
schematic of the model’s operations and parameters. Each cell operates independently of the
others, and each can force a new vessel to sprout from the network when its
distance to the network exceeds some threshold (which is directly related to
metabolic activity or oxygen consumption).
Growth of the embedded vascular network is strictly reactive and space
driven, rather than being autonomous and network driven. Left lower. An
illustration of the model’s angiogenesis during 8 iterations. In embryonic conditions, cells multiply and
the host space enlarges, and the vessels keep up with the host, maintaining a
tightly controlled vascular density.
For illustration purposes, the space or tissue has been scaled down with each iteration so that each panel is the same size,
the vessels appearing to get denser with each iteration. A space-dividing system with a
space-filling network, and a space multiplying
system with an isodensity network are identical under simple scalar
normalization, and from a dynamical and topological point of view they are
identical. While normal embryogenesis
involves a density invariant system expanding into a multiplying space,
Integra histogenesis is interesting in that it involves a space of fixed
volume which subdivides into closer control points as regenerating clusters
fill space and grow to confluence. The
model describes both situations equivalently. Middle and right lower.
The model plots images of calculated vascular networks. They are shown here juxtaposed against
blood vessels of various organs, tissues, and species, and also veins from
plant leaves. Note that there are many
types of vascular morphology, but they are all equivalent in that just one
single dynamical process creates them all, described by three parameters
representing the diffusion and reactions of oxygen, angiogenic cytokines,
host cells, and angioblasts. Right upper.
Mathematically speaking, vascular networks are fractal r-nets, fractal
being a form of geometry in which similar morphologies repeat at different
scales due to the repetitive application of a recursive dynamical process,
and an r-net being a topological entity in which no point in a host space is
more than distance r from an embedded network. While the VT output (lower panel) may look elaborate and
complex, it is actually very efficient and economical. If a simple geometric lattice was drawn on
the same space (upper panel), using the same r-value (the ischemia
threshold), in the way an engineer might design a distribution network, and
the lengths of the two networks are compared, the VT network is only 10%
longer than the idealized network.
This economy, known in biological and other complex systems as
“parsimonious self organization” means that, for blood vessels, the body
never makes any more blood vessels than just what is needed to supply the
logistical needs of the host tissue.
The consequences of this can be seen in the next slide. |
|
|
|
|
SLIDE 63Inflammatory wound healing has an open loop interaction
between stimulatory macrophages and responder angioblasts. Revascularization does not suppress the
macrophages, and they keep attracting new vessels, until there is a dense
proliferation of new vessels, far in excess of what is normally needed by
healthy tissues. This can be seen
histologically as an excessive “unnatural” number of unusually large blood
vessels, and it is seen grossly as color saturated bright red “granulation
tissue”, the clinical signature of inflammatory wound healing, due to
excessive blood volume in the excessive network. In contrast, Integra histogenesis results in a controlled
angiogenesis, in which there is feedback between burgeoning histiogenic cells
and the vessels that they attract, the same kind of
closed loop control that governs efficient embryonic vasculogenesis. The consequence is that Integra, just like
normal tissues, has only just exactly the right number of blood vessels
needed for normal development and function of the host tissue. Regenerated Integra is like any other
normal fibrous tissue such as dermis and muscular fascias, white or only
marginally pink, because blood volume in these tissues is slight. Left, inflammatory repair.
Under the microscope, angiogenesis is seen as dense, closely
distributed large vessels carrying large volumes of blood. This high blood density is seen grossly as
the exuberant red color of granulation tissue. Right, Integra histogenesis. Microscopically, vessel density is low,
comparable to normal dermis and fascias, and vessel caliber is small and more
uniform, capillary and pre- and post-capillary sizes. Grossly, the lower density of vessels and
blood means that the regenerated tissue appears pale. The regenerated material is sufficiently thin to be slightly
translucent, so the gross color appears in part like whatever is behind the
thin material, lumbar muscles in the middle photo, and achilles tendon in the
lower photo. While embryonic histogenesis is not explicitly
illustrated, its similarity to Integra histogenesis can be inferred. Since this discussion is focused on blood
vessels, the VT model was used as a way to illustrate how embryonic
development has reference-controlled vasculogenesis which leads to a normal
density of blood vessels. Tissues with
low metabolic requirements are pale due to a relatively low density of blood
vessels, and direct clinical observation of any normal dermis or fascia
confirms this. Regenerated Integra,
which is structurally and functionally similar to these tissues, looks the
same as them, and quite distinct from normally high metabolic and densely
vascularized or abnormally hypervascular tissues such as liver, kidney, and
wound module granulation. |
|
|
|
|
SLIDE 64Another approach to showing how Integra histogenesis and
embryonic histogenesis are similar to each other and distinct from inflammatory
repair is to focus on the fibrous cells and their progenitors. Left, inflammatory repair.
Top, the macrophage zone of the normal wound, with streaming
angioblasts leading the way into the area of chemotactic stimulation. Fibro-progenitor cells are yet to
appear. Middle, fibrous progenitor
cells are appearing between new blood vessels. The discrimination between histioblast and
fibroblast is moot, because they are the same, in earlier or later phases of
function and maturation, “fibro-histioblasts”. At this stage, these cells are young,
relatively large, round, and fairly dense or numerous. Young collagen is appearing. A close up view, bottom, shows that these
cells remain highly individualized, with discrete or distinct somas,
separated by GAG or collagen matrix.
These features become more exaggerated as the tissue matures, the
cells getting sparser and flatter as collagen becomes denser and
thicker. Center and right, Integra histogenesis. The earliest cells are already in the
fibroblast lineage. In a normal wound,
the earliest cells are the non-native (blood borne) non-fibrogenic
macrophages. They are under the
control of inflammatory mediators, and they then become the agents of control
for the subsequent repair process. In
Integra, the earliest cells are locally derived, they are under the control
of the matrix (presumably the aminoglycan), and they are themselves
fibrogenic and destined to become part of the tissue. They too further regulate histogenesis, but
passively, by simply trying to do their proliferative mitotic and fibrogenic
job, and attracting circulation as needed, according to principles of
embryonic vasculogenesis. Center top
and middle, the earliest cells are seen in transition to syncytial
histioblasts. Note that unlike
macrophages which must make a de novo matrix of GAG’s for themselves and
subsequent responder cells, these cells alight on the collagen-GAG matrix of
the Integra and then start to do their job.
Center bottom, as these cells proliferate, they develop features
distinct from the early fibroblasts of inflammatory repair. Note the mitosis. In comparison to macrophages, there is no
comparison, because macrophages are non-mitotic. In comparison to normal fibro-histioblasts,
these cells are not just streaming from underlying progenitors, but once in
place, they themselves start to make new cells. Note that, unlike inflammatory
fibro-histioblasts, these cells do not have, with light microscopy, distinct
identities. They seem to form a
continuum of cytoplasm, only marginally interspersed with some fluids, ground
substance, and early proteins, i.e. they appear syncytial. Finally, note that unlike macrophages which
make no collagen, these cells are already making collagen. In comparison to inflammatory fibrohistioblasts,
this early collagen is very gossamer, and relatively scant in comparison to
the cytoplasm which takes up most of the space. Right middle and bottom, other views of
these syncytial histioblasts and their effects. The syncytial-like clustering of cells is
apparent. Even as these cells start to
make denser collagen, it remains very amorphous and non-fibrous in
appearance. Right top, embryonic features. While embryonic histogenesis is not
directly illustrated here, attention is drawn to published
sources that have studied this subject.
In the paper quoted (“Ultrastructural Aspects of Human Skin During the
Embryonic, Fetal, Premature, Neonatal, and Adult Periods of Life”, Holbrook
KA and Smith LT, Birth Defects, v.17-2: pp.9 -38, 1981) the histology of
human dermal embryogenesis is described in detail, including this quote: “The dermis of the 1-2 month old embryo is a watery
cellular network of mesenchyme that is joined, through long slender
pseudopodia processes and specialized intercellular junctions, into a
syncytium. The undifferentiated
mesenchymal cells at first secrete a matrix that is primarily ground
substance . . . argyrophilic fibrils in the 20-30 nm range are visible at the
light microscope level. . . In the 3rd month, cells separate as the dermis
becomes richer in fibrous components.
Continued deposition of fibers and separation of cells have been
referred to as a ripening of the connective tissue.
. . in the fetal dermis of 14-21 weeks [4-5 months] fibroblasts have assumed
a typical spindle shape and no longer retain cell-to-cell contacts.” This description parallels exactly what is seen in these
early stages of Integra histogenesis.
Using electron microscopy, the individuality of these cells can be
identified, but they are very tightly associated, appearing as a syncytium
with light microscopy. The Holbrook
& Smith paper is the source of the term “syncytial” histioblast opted for
in this study. |
|
|
|
|
SLIDE 65This is a review, a side by side comparison of
inflammatory wound healing, Integra histogenesis, and embryonic histogenesis,
focusing on certain features of these processes. In inflammatory repair, injury to mature tissues triggers
inflammation which then leads to inflammatory repair. In Integra, inflammation not only does not
occur, but the early normal inflammatory response to the surgical wound is
suppressed. Normal embryonic
development does not of course develop against a background of inflammation,
but what is interesting is what happens after a fetal wound. Fetal wound healing studies show that an
intrauterine fetal injury or wound does not trigger inflammation. The injury simply repairs itself by the
accumulation of new cells and GAGs and a resumption of tissue specific
histogenesis. With inflammation, wound repair is triggered by extrinsic
marrow-derived cells (monocytes-macrophages) which do not themselves become
part of the healed scar. With Integra
tissue regeneration and embryonic histogenesis, the repair process is triggered
and executed by local mesenchymal cells which will ultimately become part of
the tissue. In a normal wound, angioblasts and new blood vessels are
the first cells and structures which are derived from local mesenchymal stem
sources and which will become part of the final tissue. Only after the vessels are in place can
fibroplastic cells appear and function.
In Integra, the first local histogenetic cells are the syncytial
histioblasts, which function autonomously as young fibroblasts. Blood vessels appear secondarily, only as
the original cells feel the need for revascularization. This order of appearance is the same as for
embryonic histogenesis, in which tissue-specific cells grow autonomously, and
vasculogenesis is a purely reactive space-driven response to their needs. In a normal wound, fibroplasia is effected
by fibro-histioblasts, small fibrous forming cells that come in behind the
new blood vessels, and which immediately start to make thick fibrous
collagen, the cells gradually maturing as they become entrapped in the
lamellar directional matrix. In
Integra, later, “second set” fibroplastic cells eventually undergo a similar
maturation after blood vessels appear, but the earliest fibroplastic cells
are the large syncytial ones, which make a very fine fibrillar or amorphous
collagen, just as occurs during the embryogenesis of the normal dermis. In a normal wound, angiogenesis is stimulated by a front
of macrophages, and angioblasts grow toward this planar target. The stimulus, the
macrophages, are extrinsic to the tissue in two ways: (1) they occupy a physical locale on the
surface or outside of the existing tissue or tissue-to-be, and (2) they are
not destined to be part of the ultimate new tissue. The angioblast-macrophage interaction is
open loop, angiogenesis failing to suppress macrophage stimulation, resulting
in unconstrained vessel growth. In
Integra histogenesis and embryonic histogenesis alike, the dynamics of
angiogenesis are completely different.
Angioplasia is stimulated by cells intrinsic to the developing tissue,
distributed internally within the space of the new tissue. The interaction of angioblasts and
stimulatory syncytial or fibroblastic cells is closed loop, revascularization
causing feedback suppression of further angiogenic stimulus, resulting in a
tightly controlled vascular density that precisely meets the metabolic needs
of the host tissue (remember the look of white Integra versus red granulation
tissue). Wound and scar contraction are hallmark features of normal
inflammatory repair. Contraction and
contractures do not occur in embryogenesis nor
Integra. Collagen bundle size,
collagen density, and orientation are likewise distinctly different, with
embryonic and Integra tissues having finer, more individualized collagen
clusters, less density, less directional orientation, and more “springiness”
(three dimensional structural compliance). In these three situations, collagen (connective proteins)
and glycosaminoglycans each have important but distinct roles. In a normal wound, GAGs must be made first,
to be the thick mucinous matrix that, in the absence of a mature tissue
model, the various cells need as a home to begin building the mature fibrous
tissue. Once inflammatory histogenesis
begins, what results is the densely collagenous structure known as scar. Collagen is the star of the show, being the
must abundant and crucial component of the system, the recognizable output,
the glue that binds the wound together.
In Integra, ground substance GAGs also appear, and collagen is
likewise a crucial end product, but the rate of appearance, distribution,
abundance, and architecture of these components is different than in a normal
wound. GAG’s appear, but only in the
limited amounts that are needed for cells to appear within given
locales. Collagen appears, but first
as a flimsy amorphous fibrillar mesh, maturing more slowly to thicker fibers,
with significant differences between this mature collagen and the thicker
denser less compliant collagen bundles of scar. Collagen is also the most abundant material
in the ultimate output of Integra, just like scar, but from an overall
physiological point of view, it is not the output but rather the input, the
original Integra matrix which is the star of the show. In inflammatory wound repair, there is no a priori
matrix. There is nothing but empty
space, and the system input, inflammation and its derivative macrophages, must make their own space and then behave as
they do. In Integra, the matrix is the
input. It is the signal
which tells incipient inflammation to subside, the signal which allows
pioneer cells to find a cozy place to hang out, and the signal which allows
pioneer cells to adhere, layer out, and begin life as large mitotic,
proteogenic, embryonic-like syncytial cells.
Collagen by itself is not known to do this. Given the central role of the sulfurated
and carboxylated GAGs (hyaluronan, chondroitin, etc.) in embryogenesis and
fetal non-inflammatory wound repair, and given the similarity of Integra
histogenesis to embryonic tissue development, the hypothesis is put forth
that it is the chondroitin-6-sulfate in the Integra matrix which is the key
to its properties. Collagen presumably
provides structural competence, and may have some physiological and
pharmacological effects, but it is the chondroitin which allows
histoprogenitor cells to see an opportunity to begin creating normal
tissue. The Integra matrix was
engineered with a reticulum or mesh size comparable to the collagen reticulum
in normal skin and fascias, and the influence of local structural geometry
and pore size on cell behavior is very well studied (cf bone graft materials
which are engineered or selected because of their similarity to the size of
haversian canals). Structural geometry
and mechanics and possibly other factors also confer crucial features of
Integra’s behavior, but it can be reasoned that the aminoglycan has a pivotal
role, just as the same class of aminoglycans are pivotal in embryogenesis. Recall that the overall dynamics of inflammatory repair,
being auto-amplifying and open loop, result in an
excessive accumulation of repair elements:
blood vessels, fibrous cells, and collagen. The thick, red, non-compliant scar, as seen
grossly and histologically, is testimony to this. Once the wound is “healed”. ie epithelialized, the wound remodels itself back toward a
model of normal dermis or fascia, dismantling excess materials. This process of “overshoot then involute”
starts rapidly, with the peak overshoot accumulation occurring within days to
weeks. The “involute” maturation phase
then occurs very slowly, taking months or years to complete. With Integra, there is no overshoot. As a regulated or controlled system having
feedback and references and regulators, the system gradually and
progressively builds a model of tissue which asymptotically approaches the
final result. This consolidation works
toward an anatomy which is very comparable to normal dermis, and it does so
in an intermediate time frame, building up over weeks to months. There is no formal long term maturation,
only (presumably) the gradual physiological turnover of collagen, which along
slow hydrolysis of the matrix gradually results in the final tissue looking
ever more like dermis. The anatomy
that Integra is building toward is simply a model of embryonic tissue, following
the same gradual asymptotic build of tissue that occurs during normal
embryogenesis. Even when the embryo is
injured (“fetal wound repair”), the inflammatory overshoot-then-involute
process never occurs, only the resumption of the normal evenly paced
histogenesis that creates normal tissues. |
|
|
|
|
SLIDE 66SUMMARY. In normal mature subjects, injury begets inflammation
begets inflammatory wound module repair.
In Integra, the normal inflammatory response to injury is suppressed,
and instead of inflammatory repair leading to scar, the material heals by the
gradual build of nearly normal tissue, a process which dynamically and
histologically is comparable to normal embryonic histogenesis. While these two processes share some
general similarities and parallel each other to an extent, the details of
each process are crucially different.
The properties and constituents of the Integra matrix are directly
responsible for these differences. The
physical and biological characteristics of the healed material are clinically
superior to scar and customary skin grafts, approaching the quality of normal
skin, as might be expected given the similarities of Integra and embryonic
histogenesis, and their distinction from inflammatory repair. |
END
Histogenesis versus Wound Repair: the Anatomy of Integra’s Properties
Marc
E. Gottlieb, MD, FACS |